Armodafinil as Adjunctive Therapy in Adults With Cognitive Deficits Associated With Schizophrenia: A 4-Week, Double-Blind, Placebo-Controlled Study
Objective: To evaluate the efficacy and tolerability of armodafinil, the longer-lasting isomer of modafinil, as adjunctive therapy in patients with schizophrenia.
Method: This 4-week, randomized, double-blind, placebo-controlled, proof-of-concept study was conducted between July and December 2007. Patients had a history of stable schizophrenia (DSM-IV-TR criteria) for ≥ 8 weeks and were treated with oral risperidone, olanzapine, or paliperidone for ≥ 6 weeks at stable doses for ≥ 4 weeks. Patients were randomly assigned to once-daily placebo or armodafinil 50, 100, or 200 mg. The primary efficacy measure was the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery. Secondary outcome measures included the Positive and Negative Syndrome Scale (PANSS) and the Scale for Assessment of Negative Symptoms (SANS).
Results: Sixty patients were randomly assigned (15 in each group). No apparent differences between groups in the MATRICS composite score were observed (mean ± SD change from baseline to final visit: armodafinil 50 mg, 1.9 ± 6.22; 100 mg, 2.8 ± 7.98; 200 mg, 2.9 ± 4.72; placebo, 2.2 ± 5.06). The mean ± SD changes in PANSS total scores were -6.3 ± 7.25 for armodafinil 200 mg and -1.7 ± 4.89 for placebo at final visit (effect size = 0.73; 95% CI, -0.08 to 1.54) and PANSS negative symptoms scores were -3.4 ± 2.07 and 0.1 ± 1.93 (effect size = 1.69; 95% CI, 0.78 to 2.60), respectively. Although reductions in SANS total score were observed with both armodafinil and placebo at final visit, no between-group difference was shown. Armodafinil was generally well tolerated, with diarrhea and headache the most commonly reported adverse events. There was no evidence of worsening of psychosis with adjunctive armodafinil.
Conclusions: In this 4-week study, adjunctive armodafinil was not associated with an improvement in cognitive measures, but armodafinil 200 mg/d appeared to mitigate the negative symptoms of schizophrenia. Treatment was generally well tolerated.
Trial Registration: clinicaltrials.gov Identifier: NCT00487942.
J Clin Psychiatry 2010;71(11):1475-1481
© Copyright 2010 Physicians Postgraduate Press, Inc.
Submitted: December 30, 2009; accepted May 20, 2010.
Online ahead of print: August 24, 2010 (doi:10.4088/JCP.09m05950gry).
Corresponding author: John M. Kane, MD, Department of Psychiatry, The Zucker Hillside Hospital, Glen Oaks, NY 11004-1150 ([email protected]).
Schizophrenia is a chronic, severely debilitating psychiatric disorder that affects up to 1% of the population worldwide.1 Characteristics of schizophrenia include overt psychotic, ie, positive, symptoms, such as hallucinations and delusions, as well as negative symptoms, such as social withdrawal and loss of drive.2 Although currently available antipsychotic medications generally mitigate the positive symptoms of schizophrenia, they are not effective for the negative symptoms of some patients, leading to continuing disability.3-6
Modafinil, a racemic compound containing R-modafinil and S-modafinil, has been investigated as a treatment for sedation associated with antipsychotic medications7 because it has less potential for abuse and is associated with fewer peripheral and central adverse events compared with amphetamines.8,9 Modafinil has also been evaluated for its effects on negative symptoms and cognitive deficits in schizophrenia, with mixed results. In randomized, placebo-controlled, repeated-dose studies in patients with schizophrenia, modafinil treatment significantly improved short-term verbal memory span and attentional set shifting,10 and it increased functional magnetic resonance imaging activation in the anterior cingulate cortex during the working memory task, which was associated with improved cognition in some patients.11 A randomized, placebo-controlled crossover study found that a single dose of modafinil decreased motor avolitional behavior as measured by actigraphy,12 and an open-label, 4-week study also showed that modafinil significantly improved global functioning, overall clinical condition, and fatigue, while tending to improve cognitive functioning scores, in patients with schizophrenia.13 However, in two 8-week, randomized, placebo-controlled studies in patients with schizophrenia, treatment with adjunctive modafinil did not demonstrate an effect on negative symptoms, and did not improve a range of cognitive measures, including Continuous Performance Test, Letter-Number Span, Oculomotor Delayed Response, Delayed Match to Sample Task, Controlled Oral Word Association Test, and Rey Auditory Verbal Learning Test.14,15 Overall, the efficacy of modafinil for treatment of cognitive symptoms in patients with schizophrenia remains unclear.
Armodafinil, a nonamphetamine, wakefulness-promoting medication, is the longer-lasting isomer of modafinil. Clinical studies have shown that it improves wakefulness throughout the day and aspects of memory and attention in patients with excessive sleepiness associated with treated obstructive sleep apnea, shift work disorder, or narcolepsy.16-19 The objective of this proof-of-concept study was to evaluate the efficacy and tolerability of armodafinil for cognitive deficits in adults with schizophrenia as an adjunctive agent to oral risperidone, olanzapine, or paliperidone therapy, and thereby generate hypotheses for future studies.
METHOD
Study Design and Patients
This was a randomized, double-blind, placebo-controlled, parallel-group, proof-of-concept study conducted between July and December 2007 at 11 centers in the United States. The study design included a screening period of at least 1 week, a 4-week double-blind treatment period, and a 1-week follow-up period. Efficacy and tolerability assessments were performed at clinic visits at baseline and weeks 1, 2, and 4 of treatment.
The study protocol was approved by the Independent Ethics Committee or Institutional Review Board (according to national or local regulations). The study was conducted in full accordance with the Good Clinical Practice Consolidated Guideline approved by the International Conference on Harmonisation20 and any applicable national and local laws and regulations. Written informed consent was obtained from each patient before he or she underwent any study procedures.
Eligible patients were aged 18-60 years; had a diagnosis of schizophrenia as determined by the Structured Clinical Interview for DSM-IV-TR2; were clinically stable in a nonacute phase of their illness for at least 8 weeks before the baseline visit; were being treated with oral risperidone, olanzapine, or paliperidone for at least 6 weeks before screening; and were receiving a stable dose of these antipsychotic medications for at least 4 weeks prior to screening. Patients meeting any of the following criteria were excluded from the study: Wide Range Achievement Test, Fourth Edition,21 reading subtest raw score ≤ 36; Calgary Depression Scale for Schizophrenia (CDSS)22 suicide item (item 8) score ≥ 2 (moderate or worse); moderate-to-severe depressive symptoms (CDSS score ≥ 11); any positive symptom subscale item score ≥ 4 (moderate or worse) on the Positive and Negative Syndrome Scale for Schizophrenia (PANSS),23 a clinician-rated instrument of the severity of psychopathology; modified Simpson-Angus Scale24 score for extrapyramidal side effects ≥ 7 (moderate or worse); Barnes Akathisia Scale25 global score ≥ 2; presence of tardive dyskinesia or other movement disorder; history or current active suicidal ideation or imminent risk of self-harm; diagnosis of any other axis I disorder, alcohol or substance abuse, or dependence within the previous 6 months.
Patients were randomly assigned (1:1:1:1) to receive armodafinil 50, 100, or 200 mg/d or placebo. Patients took 4 tablets once daily in the morning. The armodafinil or matching placebo dose was initiated at 50 mg/d and was titrated up by 50 mg/d on days 2, 4, and 6, as applicable, to the randomized dose.
Study Assessments
The primary efficacy measure was the mean change from baseline at final visit (last observation, ie, week 4 or early termination) in the composite score on the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery. The MATRICS Consensus Cognitive Battery contains 10 tests to measure cognitive performance in 7 domains of cognition: speed of processing, attention/vigilance, working memory (nonverbal and verbal), verbal learning, visual learning, reasoning and problem solving, and social cognition.26-28 Secondary efficacy measures included mean change from baseline to final visit in the following: the 7 individual MATRICS domain scores; ratings on the Clinical Global Impressions-Severity of Illness scale (CGI-S),29 a standardized, clinician-rated assessment of the severity of illness of the patient, at weeks 1, 2, and 4 or at final visit; the Scale for the Assessment of Negative Symptoms (SANS),30 a clinician-rated instrument used to rate the severity of negative symptoms of schizophrenia; scores on the PANSS, which includes 7 items measuring positive symptoms (this subscale was assessed as a safety measure), 7 items measuring negative symptoms, and 16 items forming the general psychopathology subscale; and the Epworth Sleepiness Scale (ESS), a subjective measure of the patient’s propensity for daytime sleepiness.31
Tolerability measures were determined by reports of adverse events and results of clinical laboratory tests, vital signs, modified Simpson-Angus Scale, Barnes Akathisia Scale, CDSS, PANSS, and actigraphy data related to sleep.
Statistical Analysis
The safety analysis set included all patients who received at least 1 dose of study drug, and the efficacy analysis set included all patients in the safety analysis set who had at least 1 postbaseline assessment on the MATRICS Consensus Cognitive Battery.
Patient demographic characteristics were summarized using descriptive statistics. For continuous variables, descriptive statistics were provided; for categorical variables, patient counts and percentages were provided. This study was not powered to detect significant differences between the armodafinil and placebo groups. For the primary efficacy variable, effect sizes and 95% confidence intervals (CIs) of the effect sizes for the primary efficacy measure were provided for each armodafinil treatment group (compared with the placebo group). Actual values and changes from baseline to final visit (4-week or last postbaseline measurement) were summarized using descriptive statistics. The final visit analyses were performed using the last postbaseline observation carried forward. All continuous secondary efficacy variables, at each visit and final visit, were summarized as specified for the primary efficacy variable. The categorical secondary efficacy variables of CGI-S ratings were summarized using descriptive statistics.
RESULTS
Patient Disposition and Characteristics
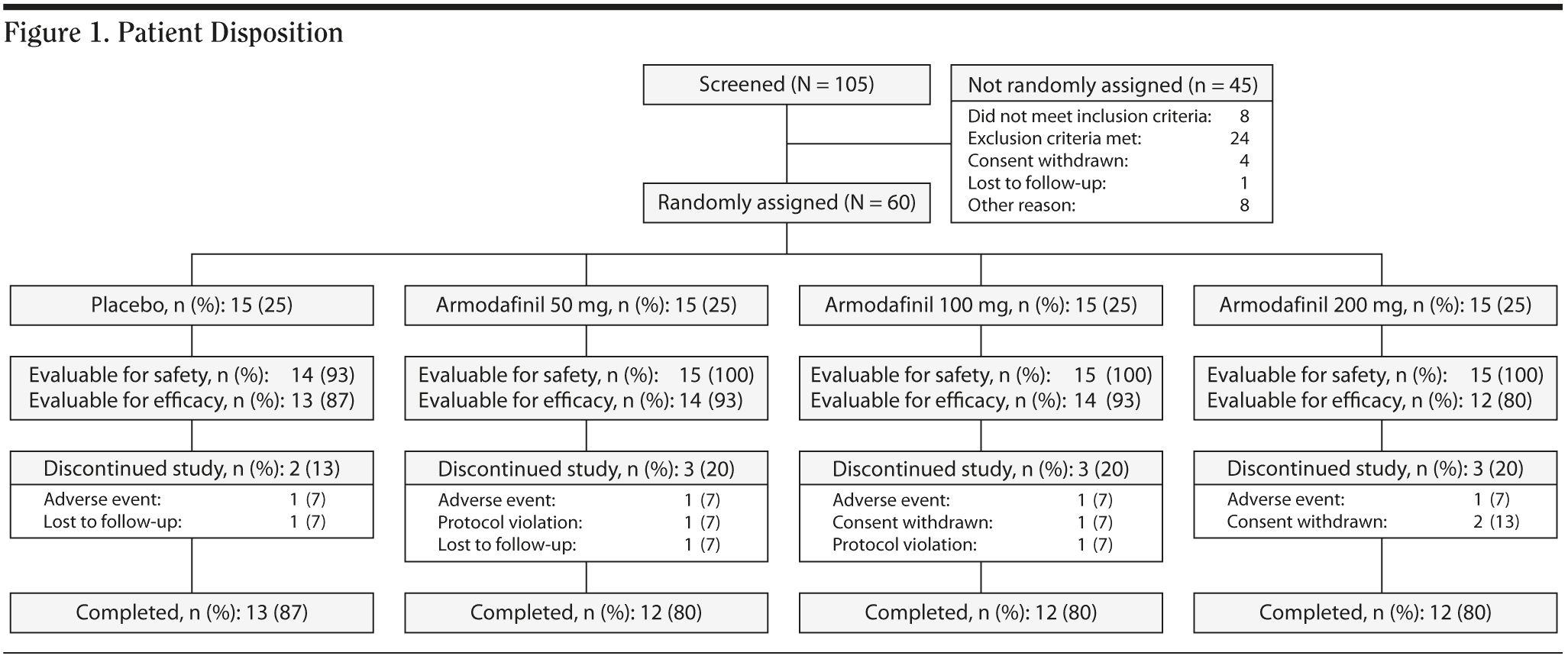
Of 105 patients screened, 60 were randomly assigned to receive armodafinil 50 mg/d (n = 15), 100 mg/d (n = 15), 200 mg/d (n = 15), or placebo (n = 15); a total of 49 patients (82%) completed the 4-week study (Figure 1). The most frequent reason for withdrawal from the study was adverse events (n = 4; n = 1 for each of the armodafinil groups and for the placebo group). The mean age of patients was 43 years; 60% (n = 36) were black, and 73% (n = 44) were men. Baseline characteristics were generally similar across the treatment groups (Table 1).
Effect of Armodafinil
Changes from baseline to final visit in cognitive deficits were similar following armodafinil or placebo on the MATRICS composite score (Table 2). No apparent differences between the armodafinil and placebo groups were observed in mean changes from baseline to final visit in any of the 7 individual MATRICS domain scores (Table 3). Mean ± SD changes from baseline to final visit in CGI-S scores were -0.07 ± 0.27 following armodafinil 50 mg, 0.07 ± 0.47 following armodafinil 100 mg, -0.17 ± 0.39 following armodafinil 200 mg, and no change with placebo. Reductions in the SANS total score were observed following armodafinil and placebo at final visit but were not considered clinically meaningful (Figure 2).
There was a greater reduction in PANSS negative symptoms subscale score in the armodafinil 200 mg group compared with the placebo group (effect size = 1.69 at final visit; 95% CI, 0.78 to 2.60), without worsening of positive symptoms (Figure 3). The reduction in the mean PANSS negative scale score was apparent by week 1 in the armodafinil 200 mg group (effect size = 0.89 at week 1; 95% CI, 0.05 to 1.74). The PANSS total score showed greater improvement in the armodafinil 200 mg group (-6.3) compared with the placebo group (-1.7) (effect size = 0.73 at final visit; 95% CI, -0.08 to 1.54). There were no clinically relevant changes in the general psychopathology subscale score: mean ± SD changes from baseline to final visit were -1.5 ± 4.8 following armodafinil 50 mg, -0.7 ± 4.1 following armodafinil 100 mg, -2.1 ± 3.7 following armodafinil 200 mg, and -0.9 ± 3.1 following placebo.
Mean changes in ESS score from baseline to final visit were -0.5 following placebo, -2.1 following armodafinil 50 mg (effect size = 0.25; 95% CI, -0.51 to 1.01), -0.6 following armodafinil 100 mg (effect size = 0.03; 95% CI, -0.73 to 0.78), and 1.0 following armodafinil 200 mg (effect size = -0.23; 95% CI, -1.01 to 0.56).
Tolerability
Armodafinil was generally well tolerated; diarrhea and headache were the most commonly reported adverse events (Table 4). One patient in each group discontinued treatment because of an adverse event, including psychotic disorder (placebo), folliculitis (armodafinil 50 mg), hostility (armodafinil 100 mg), and restlessness (armodafinil 200 mg). One adverse event was classified as serious: worsening psychosis in 1 patient in the placebo group. Psychiatric adverse events included insomnia (2 in the placebo group, and 1 each in the armodafinil 50 and 200 mg groups), restlessness (3 in the armodafinil 200 mg group), and hostility (1 patient in the armodafinil 100 mg group). No deaths occurred during the study.
The nighttime actigraphy data indicated that, in patients receiving armodafinil 200 mg compared with placebo, mean ± SD values of sleep latency increased (by 9.8 ± 26.6 min following armodafinil 200 mg vs -1.4 ± 11.0 following placebo) and mean ± SD values of sleep efficiency and total sleep time decreased (by -5.5% ± 8.8 versus 2.3% ± 6.0 and -39.9 ± 57.3 min vs 22.7 ± 107.2 min, respectively); no other differences were seen between the armodafinil and placebo groups (Table 5).
Mean changes from baseline to final visit in heart rate and diastolic blood pressure values were slightly greater among patients treated with armodafinil 200 mg (1.9 beats per min and 4.1 mm Hg, respectively) than with placebo (-3.2 beats per min and -0.6 mm Hg, respectively), but these were not considered to be clinically meaningful by investigators.
There was no worsening of symptoms as assessed by Simpson-Angus Scale, Barnes Akathisia Scale, CDSS, or PANSS positive scale score (Figure 3C); no notable differences were observed regarding laboratory values or electrocardiograms (data not shown).
DISCUSSION
In this 4-week study, adjunctive armodafinil had no apparent beneficial effect on cognitive measures in patients with schizophrenia. Changes from baseline to final visit in the MATRICS composite score were similar in the armodafinil and placebo groups. Armodafinil 200 mg/d, however, did appear to improve the negative symptoms of schizophrenia, as indicated by a reduction from baseline to final visit compared with placebo in the mean PANSS negative symptoms scale score (-3.4 versus 0.1; 95% CI, 0.78 to 2.60). The difference in the negative subscale score, combined with the similarity of changes on the positive and general psychopathology subscale scores between the 200 mg/d and placebo treatment groups, drove a reduction from baseline to final visit compared with placebo in mean PANSS total score (-6.3 vs -1.7; 95% CI, -0.08 to 1.54). Armodafinil was generally well tolerated.
Treating negative symptoms of schizophrenia is important because these symptoms are debilitating for patients and because antipsychotic therapies are often not adequate to treat them.32-34 In 2 double-blind, placebo-controlled studies, there were no significant differences in the SANS scores of patients administered modafinil and those given placebo.14,15 In the current study, armodafinil also did not appear to improve SANS scores, compared with placebo. However, armodafinil 200 mg/d improved negative symptoms from week 1 as assessed by the PANSS negative symptoms scale score.
The precise mechanisms of action of armodafinil and its effect on the neuronal pathways involved in schizophrenia are still unclear. Dopamine plays a role in the mechanism of action of the racemic mixture modafinil and, presumably, of armodafinil. Modafinil binds to the dopamine transporter (DAT),35-37 with less affinity than do methylphenidate, benztropine, and bupropion,38 and its activity at the DAT is necessary for its wakefulness-promoting effect.39 Modafinil increases extracellular levels of dopamine in the prefrontal cortex,40 and there is some evidence that it enhances activity in the prefrontal cortex in patients with schizophrenia.41 Weinberger42 proposed that underactivity of the dopaminergic connections to the prefrontal cortex, the mesocortical pathway, is the primary lesion in schizophrenia and is associated with negative symptoms. Decreased dopaminergic activity in the prefrontal cortex results in disinhibition of the mesolimbic function, which produces positive symptoms.42 A weak DAT inhibitor such as armodafinil, which may preferentially influence the dopaminergic activity in the prefrontal cortex but not in the limbic system, could, hypothetically, reduce negative symptoms without worsening positive symptoms—the effect suggested in this study.
A limitation of the current study is that it was not powered to detect differences in effect between the armodafinil and placebo groups, and so inferential statistics were not performed to compare differences in effect between study groups. The relatively small sample size (N = 60) also limits the application of these results to the general population of patients with schizophrenia in clinical practice. Although an improvement in negative symptoms following armodafinil 200 mg/d was indicated by the PANSS negative symptom score, there was no apparent improvement in SANS score. According to an analysis comparing the PANSS negative symptoms subscale and SANS scores in a sample of patients with schizophrenia, the total scores were highly correlated with each other, but the 2 scales predicted the various cognitive, motivational, and social dimensions of the negative syndrome to different degrees.43 Together, these limitations point to the need for further characterization of armodafinil’s effects in larger, adequately powered studies, involving populations of patients with schizophrenia defined in terms of baseline cognitive functioning and degree and type of negative symptoms. Future studies may also investigate whether changes in measures of cognitive functioning are accompanied by improvements in patients’ quality of life or daily functioning.
The lack of a consensus cognitive battery has been a major barrier to the evaluation of the cognitive effects of new treatments in schizophrenia. The National Institute of Mental Health developed the MATRICS initiative, a consensus cognitive battery for use as a research tool in clinical studies in schizophrenia.28,44 This study adds to the small but growing number of published studies to use the MATRICS Consensus Cognitive Battery to assess cognition in patients with schizophrenia.
CONCLUSION
In this proof-of-concept study, daily armodafinil 50 mg, 100 mg, or 200 mg, administered over 4 weeks as adjunctive treatment to oral risperidone, olanzapine, or paliperidone, was not associated with any apparent improvement in cognitive deficits in patients with schizophrenia as assessed by the MATRICS Consensus Cognitive Battery. Adjunctive armodafinil was generally well tolerated, with no evidence of worsening of psychosis. The effects of armodafinil 200 mg on negative symptoms warrant further study of the potential efficacy of this drug in their treatment.
Drug names: armodafinil (Nuvigil), benztropine (Cogentin and others), bupropion (Aplenzin, Wellbutrin, and others), modafinil (Provigil), olanzapine (Zyprexa), paliperidone (Invega), risperidone (Risperdal and others).
Author affiliations: Department of Psychiatry, The Zucker Hillside Hospital, Glen Oaks, New York (Dr Kane); Department of Psychiatry, Yale University School of Medicine, Schizophrenia Biological Research Center, Veteran Affairs Connecticut Healthcare System, West Haven, Connecticut (Dr D’ Souza); Department of Psychiatry and Behavioral Sciences, Duke University, Durham, North Carolina (Drs Patkar and Keefe); and Clinical Research (Drs Youakim and Tiller) and Biometrics (Dr Yang), Cephalon Inc, Frazer, Pennsylvania.
Potential conflicts of interest: Dr Kane has provided consultancy or participated in advisory boards or speakers’ bureaus for AstraZeneca, Targacept, Johnson & Johnson, Otsuka, Takeda, Intra-Cellular Therapies, and Cephalon; and has received honoraria from AstraZeneca, Eli Lilly, Bristol-Myers Squibb, and Boehringer Ingelheim. Dr D’ Souza has received research support awarded to Yale University School of Medicine from AstraZeneca, Cephalon, National Institute on Alcohol Abuse and Alcoholism, National Institute on Drug Abuse, National Institute of Mental Health, Organon/Schering-Plough, Pfizer, sanofi-aventis, Stanley Medical Research Institute, and Veterans Affairs Research and Development. Dr Patkar has received grant/research support from Alkermes, Bristol-Myers Squibb, Cephalon, Forest, GlaxoSmithKline, Merck, Lundbeck, Jazz, Pfizer, Titan, and Shire; has been a consultant to Bristol-Myers Squibb, Reckitt Benckiser, and Forest; has served on speakers/advisory boards for or has received honoraria from Alkermes, Bristol-Myers Squibb, Merck, Pfizer, and Reckitt Benckiser. Drs Youakim and Yang are employees and stock shareholders of Cephalon. Dr Tiller is an employee and stock shareholder of Cephalon; Dr Tiller’s spouse is a stock shareholder of GlaxoSmithKline. Dr Keefe has been a consultant to Abbott, BioLineRx, Dainippon Sumitomo, Eli Lilly, Johnson & Johnson, Lundbeck, Memory Pharmaceuticals, Merck, NeuroSearch, Orion, Otsuka, Pfizer, Roche, sanofi-aventis, Wyeth, and Xenoport; has received honoraria from AstraZeneca and Bristol-Myers Squibb; has received unrestricted educational funding from AstraZeneca; has received funding for travel from Cephalon; has served on advisory boards for Eli Lilly and Wyeth; has been a speaker for Eli Lilly; and has received royalties from Brief Assessment of Cognition in Schizophrenia (BACS) and Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Battery (BACS Symbol Coding).
Funding/support: This study was sponsored by Cephalon, Inc, Frazer, Pennsylvania.
Previous presentations: This study was presented in part at the American College of Neuropsychopharmacology 47th Annual Meeting, December 7-11, 2008, Scottsdale, Arizona; 2009 International Congress on Schizophrenia Research, March 28-April 1, 2009, San Diego, California; the 49th New Clinical Drug Evaluation Unit (NCDEU) Annual Meeting, June 29-July 2, 2009, Fort Lauderdale, Florida; the 162nd Annual Meeting of the American Psychiatric Association, May 16-21, 2009, San Francisco, California; and the 61st Annual Meeting of the American Academy of Neurology, April 25-May 2, 2009, Seattle, Washington.
Acknowledgments: Editorial support was provided by Michelle Hughes, BSc (Chesire, United Kingdom) and additional support was provided by the Curry Rockefeller Group, LLC (Tarrytown, New York). This editorial support included assistance in literature searches, editing and fact checking, graphic support, and, at the request of and with guidance from the authors, support for the preparation of the outline and drafting of this manuscript, and was funded by Cephalon, Inc.
REFERENCES
1. Freedman R. Schizophrenia. N Engl J Med. 2003;349(18):1738-1749. PubMed doi:10.1056/NEJMra035458
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC: American Psychiatric Association; 2000.
3. Buchanan RW. Persistent negative symptoms in schizophrenia: an overview. Schizophr Bull. 2007;33(4):1013-1022. PubMed doi:10.1093/schbul/sbl057
4. Goldberg TE, Greenberg RD, Griffin SJ, et al. The effect of clozapine on cognition and psychiatric symptoms in patients with schizophrenia. Br J Psychiatry. 1993;162(1):43-48. PubMed doi:10.1192/bjp.162.1.43
5. Keefe RS, Bilder RM, Davis SM, et al. Neurocognitive Working Group. Neurocognitive effects of antipsychotic medications in patients with chronic schizophrenia in the CATIE Trial. Arch Gen Psychiatry. 2007;64(6):633-647. PubMed doi:10.1001/archpsyc.64.6.633
6. Keefe RS, Silva SG, Perkins DO, et al. The effects of atypical antipsychotic drugs on neurocognitive impairment in schizophrenia: a review and meta-analysis. Schizophr Bull. 1999;25(2):201-222. PubMed
7. Makela EH, Miller K, Cutlip WD 2nd. Three case reports of modafinil use in treating sedation induced by antipsychotic medications. J Clin Psychiatry. 2003;64(4):485-486. PubMed doi:10.4088/JCP.v64n0420h
8. Jasinski DR. An evaluation of the abuse potential of modafinil using methylphenidate as a reference. J Psychopharmacol. 2000;14(1):53-60. PubMed doi:10.1177/026988110001400107
9. Jasinski DR, Kovacević-Ristanović R. Evaluation of the abuse liability of modafinil and other drugs for excessive daytime sleepiness associated with narcolepsy. Clin Neuropharmacol. 2000;23(3):149-156. PubMed doi:10.1097/00002826-200005000-00004
10. Turner DC, Clark L, Pomarol-Clotet E, et al. Modafinil improves cognition and attentional set shifting in patients with chronic schizophrenia. Neuropsychopharmacology. 2004;29(7):1363-1373. PubMed doi:10.1038/sj.npp.1300457
11. Spence SA, Green RD, Wilkinson ID, et al. Modafinil modulates anterior cingulate function in chronic schizophrenia. Br J Psychiatry. 2005;187(1):55-61. PubMed doi:10.1192/bjp.187.1.55
12. Farrow TF, Hunter MD, Haque R, et al. Modafinil and unconstrained motor activity in schizophrenia: double-blind crossover placebo-controlled trial. Br J Psychiatry. 2006;189(5):461-462. PubMed doi:10.1192/bjp.bp.105.017335
13. Rosenthal MH, Bryant SL. Benefits of adjunct modafinil in an open-label, pilot study in patients with schizophrenia. Clin Neuropharmacol. 2004;27(1):38-43. PubMed doi:10.1097/00002826-200401000-00011
14. Pierre JM, Peloian JH, Wirshing DA, et al. A randomized, double-blind, placebo-controlled trial of modafinil for negative symptoms in schizophrenia. J Clin Psychiatry. 2007;68(5):705-710. PubMed doi:10.4088/JCP.v68n0507
15. Sevy S, Rosenthal MH, Alvir J, et al. Double-blind, placebo-controlled study of modafinil for fatigue and cognition in schizophrenia patients treated with psychotropic medications. J Clin Psychiatry. 2005;66(7):839-843. PubMed doi:10.4088/JCP.v66n0705
16. Czeisler CA, Walsh JK, Wesnes KA, et al. Armodafinil for treatment of excessive sleepiness associated with shift work disorder: a randomized controlled study. Mayo Clin Proc. 2009;84(11):958-972. PubMed doi:10.4065/84.11.958
17. Harsh JR, Hayduk R, Rosenberg R, et al. The efficacy and safety of armodafinil as treatment for adults with excessive sleepiness associated with narcolepsy. Curr Med Res Opin. 2006;22(4):761-774. PubMed doi:10.1185/030079906X100050
18. Roth T, Rippon GA, Arora S. Armodafinil improves wakefulness and long-term episodic memory in nCPAP-adherent patients with excessive sleepiness associated with obstructive sleep apnea. Sleep Breath. 2008;12(1):53-62. PubMed doi:10.1007/s11325-007-0137-7
19. Roth T, White D, Schmidt-Nowara W, et al. Effects of armodafinil in the treatment of residual excessive sleepiness associated with obstructive sleep apnea/hypopnea syndrome: a 12-week, multicenter, double-blind, randomized, placebo-controlled study in nCPAP-adherent adults. Clin Ther. 2006;28(5):689-706. PubMed doi:10.1016/j.clinthera.2006.05.013
20. ICH harmonised tripartite guideline. Guideline for good clinical practice, E6(R1). International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. http://www.ich.org/LOB/media/MEDIA482.pdf. Updated June 10, 1996. Accessed November 20, 2009.
21. Wilkinson GS, Robertson GJ. Wide Range Achievement Test 4 Professional Manual. Lutz, FL: Psychological Assessment Resources; 2006.
22. Addington D, Addington J, Schissel B. A depression rating scale for schizophrenics. Schizophr Res. 1990;3(4):247-251. PubMed doi:10.1016/0920-9964(90)90005-R
23. Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13(2):261-276. PubMed
24. Simpson GM, Angus JW. A rating scale for extrapyramidal side effects. Acta Psychiatr Scand Suppl. 1970;45(S212):11-19. PubMed doi:10.1111/j.1600-0447.1970.tb02066.x
25. Barnes TR. A rating scale for drug-induced akathisia. Br J Psychiatry. 1989;154(5):672-676. PubMed doi:10.1192/bjp.154.5.672
26. Buchanan RW, Davis M, Goff D, et al. A summary of the FDA-NIMH-MATRICS workshop on clinical trial design for neurocognitive drugs for schizophrenia. Schizophr Bull. 2005;31(1):5-19. PubMed doi:10.1093/schbul/sbi020
27. Kern RS, Nuechterlein KH, Green MF, et al. The MATRICS Consensus Cognitive Battery, part 2: co-norming and standardization. Am J Psychiatry. 2008;165(2):214-220. PubMed doi:10.1176/appi.ajp.2007.07010043
28. Nuechterlein KH, Green MF, Kern RS, et al. The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am J Psychiatry. 2008;165(2):203-213. PubMed doi:10.1176/appi.ajp.2007.07010042
29. Guy W. ECDEU Assessment Manual for Psychopharmacology, Revised. Rockville, MD: US Department of Health and Human Services; 1976:218-222.
30. Andreasen NC. The Scale for the Assessment of Negative Symptoms (SANS): conceptual and theoretical foundations. Br J Psychiatry Suppl. 1989;(7):49-58. PubMed
31. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540-545. PubMed
32. Kopelowicz A, Zarate R, Tripodis K, et al. Differential efficacy of olanzapine for deficit and nondeficit negative symptoms in schizophrenia. Am J Psychiatry. 2000;157(6):987-993. PubMed doi:10.1176/appi.ajp.157.6.987
33. Milev P, Ho BC, Arndt S, et al. Predictive values of neurocognition and negative symptoms on functional outcome in schizophrenia: a longitudinal first-episode study with 7-year follow-up. Am J Psychiatry. 2005;162(3):495-506. PubMed doi:10.1176/appi.ajp.162.3.495
34. Rosenheck R, Dunn L, Peszke M, et al. Department of Veterans Affairs Cooperative Study Group on Clozapine in Refractory Schizophrenia. Impact of clozapine on negative symptoms and on the deficit syndrome in refractory schizophrenia. Am J Psychiatry. 1999;156(1):88-93. PubMed
35. Mignot E, Nishino S, Guilleminault C, et al. Modafinil binds to the dopamine uptake carrier site with low affinity. Sleep. 1994;17(5):436-437. PubMed
36. Volkow ND, Fowler JS, Logan J, et al. Effects of modafinil on dopamine and dopamine transporters in the male human brain: clinical implications. JAMA. 2009;301(11):1148-1154. PubMed doi:10.1001/jama.2009.351
37. Zolkowska D, Jain R, Rothman RB, et al. Evidence for the involvement of dopamine transporters in behavioral stimulant effects of modafinil. J Pharmacol Exp Ther. 2009;329(2):738-746. PubMed doi:10.1124/jpet.108.146142
38. Madras BK, Xie Z, Lin Z, et al. Modafinil occupies dopamine and norepinephrine transporters in vivo and modulates the transporters and trace amine activity in vitro. J Pharmacol Exp Ther. 2006;319(2):561-569. PubMed doi:10.1124/jpet.106.106583
39. Wisor JP, Nishino S, Sora I, et al. Dopaminergic role in stimulant-induced wakefulness. J Neurosci. 2001;21(5):1787-1794. PubMed
40. de Saint Hilaire Z, Orosco M, Rouch C, et al. Variations in extracellular monoamines in the prefrontal cortex and medial hypothalamus after modafinil administration: a microdialysis study in rats. Neuroreport. 2001;12(16):3533-3537. PubMed doi:10.1097/00001756-200111160-00032
41. Hunter MD, Ganesan V, Wilkinson ID, et al. Impact of modafinil on prefrontal executive function in schizophrenia. Am J Psychiatry. 2006;163(12):2184-2186. PubMed doi:10.1176/appi.ajp.163.12.2184
42. Weinberger DR. Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry. 1987;44(7):660-669. PubMed
43. Welham J, Stedman T, Clair A. Choosing negative symptom instruments: issues of representation and redundancy. Psychiatry Res. 1999;87(1):47-56. PubMed doi:10.1016/S0165-1781(99)00042-6
44. Marder SR, Fenton W. Measurement and Treatment Research to Improve Cognition in Schizophrenia: NIMH MATRICS initiative to support the development of agents for improving cognition in schizophrenia. Schizophr Res. 2004;72(1):5-9. PubMed doi:10.1016/j.schres.2004.09.010