This Academic Highlights section of The Journal of Clinical Psychiatry presents the highlights of the videoconference “Utilizing the Anxious Distress Specifier to Develop Treatment Strategies for Patients With Major Depressive Disorder,” which was held in June 2017.
The videoconference was chaired by Michael E. Thase, MD, Director, Mood and Anxiety Program, and Professor of Psychiatry, University of Pennsylvania, Philadelphia, Pennsylvania. The faculty were Richard H. Weisler, MD, Adjunct Professor of Psychiatry, University of North Carolina at Chapel Hill; J. Sloan Manning, MD, Adjunct Associate Professor, Department of Family Medicine at the University of North Carolina, Chapel Hill; and Madhukar H. Trivedi, MD, Professor of Psychiatry, University of Texas Southwestern Medical Center, Dallas, Texas.
Financial disclosures: Dr Thase has been an advisor/consultant for Acadia, Alkermes, Allergan (Forest, Naurex), AstraZeneca, Cerecor, Eli Lilly, Fabre-Kramer, Gerson Lehrman Group, Guidepoint Global, Johnson & Johnson, (Janssen, Ortho-McNeil), Lundbeck, MedAvante, Merck, Moksha8, Nestlש (PamLab), Novartis, Otsuka, Pfizer, Shire, Sunovion, and Takeda; has received grant support from Agency for Healthcare Research and Quality, Alkermes, AssureRx, Avanir, Forest, Janssen, Intracellular, National Institute of Mental Health, Otsuka, and Takeda; has equity holdings in MedAvante; and has received royalties from American Psychiatric Foundation, Guilford Publications, Herald House, and W. W. Norton & Company. Dr Thase’s spouse is employed by Peloton Advantage. Dr Manning has been a consultant for and served on speakers/advisory boards for Otsuka, Takeda, Lundbeck, Alkermes, and Sunovion. Dr Weisler has been a consultant to Alcobra, AltheaDx, lronshore, Lundbeck, Major League Baseball (certified MLB clinician [therapeutic use exemption]), National Football League (certified NFL clinician [therapeutic use exemption]), Neos Therapeutics, Nestlש Health Science-PamLab, Otsuka America, Rhodes, Shire, Sunovion, Supernus, and Validus; served on the speakers bureaus of Lundbeck, Neos Therapeutics, Nestlש Health Science-PamLab, Otsuka America, Rhodes, Shire, Sunovion, and Validus; and received research support from Alcobra, Allergan, Daiichi Sankyo, Genomind, Janssen, Merck, Neurim, Otsuka America, Roche-Genentech, Shire, Suven Life Sciences, and Theravance. Dr Trivedi has been a consultant to Alkermes, Allergan, Acadia, AstraZeneca, Bristol-Myers Squibb, Cerecor, Global Medical Education, Health Research Assoc, Lundbeck, Medscape, MSI Methylation Services, Merck, Naurex, Nestlש Health Science-PamLab, One Carbon Therapeutics, Otsuka America, PamLab, Pfizer, Roche, Shire Development, and Takeda; received grant/research support from National Institute of Mental Health, National Institute on Drug Abuse, Johnson & Johnson, and Janssen R&D; and received other financial/material support from Janssen R&D.
This evidence-based peer-reviewed Academic Highlights was prepared by Healthcare Global Village, Inc. Financial support for preparation and dissemination of this Academic Highlights was provided by Otsuka Pharmaceutical Development & Commercialization, Inc., and Lundbeck, Inc. The faculty acknowledges Sarah Brownd, MA, for editorial assistance in developing the manuscript. The opinions expressed herein are those of the faculty and do not necessarily reflect the views of Healthcare Global Village, Inc, the publisher, or the commercial supporters. This article is distributed by Otsuka Pharmaceutical Development & Commercialization, Inc., and Lundbeck, Inc., for educational purposes only.
The videoconference was chaired by Michael E. Thase, MD, Director, Mood and Anxiety Program, and Professor of Psychiatry, University of Pennsylvania, Philadelphia, Pennsylvania. The faculty were Richard H. Weisler, MD, Adjunct Professor of Psychiatry, University of North Carolina at Chapel Hill; J. Sloan Manning, MD, Adjunct Associate Professor, Department of Family Medicine at the University of North Carolina, Chapel Hill; and Madhukar H. Trivedi, MD, Professor of Psychiatry, University of Texas Southwestern Medical Center, Dallas, Texas.
Financial disclosures: Dr Thase has been an advisor/consultant for Acadia, Alkermes, Allergan (Forest, Naurex), AstraZeneca, Cerecor, Eli Lilly, Fabre-Kramer, Gerson Lehrman Group, Guidepoint Global, Johnson & Johnson, (Janssen, Ortho-McNeil), Lundbeck, MedAvante, Merck, Moksha8, Nestlé (PamLab), Novartis, Otsuka, Pfizer, Shire, Sunovion, and Takeda; has received grant support from Agency for Healthcare Research and Quality, Alkermes, AssureRx, Avanir, Forest, Janssen, Intracellular, National Institute of Mental Health, Otsuka, and Takeda; has equity holdings in MedAvante; and has received royalties from American Psychiatric Foundation, Guilford Publications, Herald House, and W. W. Norton & Company. Dr Thase’s spouse is employed by Peloton Advantage. Dr Manning has been a consultant for and served on speakers/advisory boards for Otsuka, Takeda, Lundbeck, Alkermes, and Sunovion. Dr Weisler has been a consultant to Alcobra, AltheaDx, lronshore, Lundbeck, Major League Baseball (certified MLB clinician [therapeutic use exemption]), National Football League (certified NFL clinician [therapeutic use exemption]), Neos Therapeutics, Nestlé Health Science-PamLab, Otsuka America, Rhodes, Shire, Sunovion, Supernus, and Validus; served on the speakers bureaus of Lundbeck, Neos Therapeutics, Nestlé Health Science-PamLab, Otsuka America, Rhodes, Shire, Sunovion, and Validus; and received research support from Alcobra, Allergan, Daiichi Sankyo, Genomind, Janssen, Merck, Neurim, Otsuka America, Roche-Genentech, Shire, Suven Life Sciences, and Theravance. Dr Trivedi has been a consultant to Alkermes, Allergan, Acadia, AstraZeneca, Bristol-Myers Squibb, Cerecor, Global Medical Education, Health Research Assoc, Lundbeck, Medscape, MSI Methylation Services, Merck, Naurex, Nestlé Health Science-PamLab, One Carbon Therapeutics, Otsuka America, PamLab, Pfizer, Roche, Shire Development, and Takeda; received grant/research support from National Institute of Mental Health, National Institute on Drug Abuse, Johnson & Johnson, and Janssen R&D; and received other financial/material support from Janssen R&D.
This evidence-based peer-reviewed Academic Highlights was prepared by Healthcare Global Village, Inc. Financial support for preparation and dissemination of this Academic Highlights was provided by Otsuka Pharmaceutical Development & Commercialization, Inc., and Lundbeck, Inc. The faculty acknowledges Sarah Brownd, MA, for editorial assistance in developing the manuscript. The opinions expressed herein are those of the faculty and do not necessarily reflect the views of Healthcare Global Village, Inc, the publisher, or the commercial supporters. This article is distributed by Otsuka Pharmaceutical Development & Commercialization, Inc., and Lundbeck, Inc., for educational purposes only.
J Clin Psychiatry 2017;78(9):1351-1362
https://doi.org/10.4088/JCP.ot17015ah1
© Copyright 2017 Physicians Postgraduate Press, Inc.
Many patients with depression also experience anxiety symptoms. For some, these symptoms reflect a coexisting, ongoing anxiety disorder, while for others, a significant surge in anxiety occurs along with—and complicates the clinical picture of—depressive episodes. The importance of anxious features that occur in the context of depressive episodes has been recognized in DSM-5 with the addition of an anxious distress specifier.
In this Academic Highlights, psychiatrists Michael E. Thase, MD; Richard H. Weisler, MD; and Madhukar H. Trivedi, MD, and primary care physician J. Sloan Manning, MD, engage in a discussion on the prevalence of anxiety in depressed patients, the emergence of the DSM-5 anxious distress specifier, characteristics associated with this symptom profile, and screening and treatment strategies.
Dr Thase began the discussion by stating that the prevalence of anxiety symptoms in MDD patients is between 40% and 60%. Gaspersz et al,1 in the Netherlands Study of Depression and Anxiety (NESDA), identified items on various self-report measures that corresponded to DSM-5 anxious distress specifier criteria and found a prevalence of 54%. In a large sample of psychiatric outpatients with MDD, Zimmerman et al2 demonstrated an even higher prevalence of patients who met specifier criteria, 78%. Further, the prevalence has been shown to be higher in primary care than in specialty care.3
The DSM-5 “with anxious distress” specifier4(p184) can be appended to depressive diagnoses and allows clinicians to both note the presence and rate the severity of anxiety symptoms. The specifier may be used for patients with at least 2 of the following symptoms during the majority of days of a major depressive episode:
Clinicians can gauge severity on the basis of the number of symptoms displayed:
- 0 or 1 symptom = no anxious distress,
- 2 symptoms = mild anxious distress,
- 3 symptoms = moderate anxious distress, and
- 4-5 symptoms = moderate to severe anxious distress (psychomotor agitation must be present).
In their overview of DSM-5 changes, Regier et al5 state that severity-based specifiers may be particularly informative because treatment for mild symptoms should differ from treatment for moderate-to-severe presentations. The specifier includes symptoms that, although they are not a part of the criteria for most mood disorders (eg, difficulty concentrating because of worry), may describe a disorder variant that causes distress and merits attention in treatment choice. Part of the DSM-5 task force’s reasoning for the addition was to allow clinicians to assign a specifier that yields information for treatment planning and tracking efforts that might have been previously obscured under a residual diagnosis of “not otherwise specified” in DSM-IV.5 “Mixed anxiety-depressive disorder” was included in DSM-IV as an area for further research, but not as an official diagnosis. Anxious depression has been defined dimensionally, syndromally, and as mixed-anxiety depressive disorder (depression and anxiety that do not meet criteria for either disorder).6 The specifier reflects a shift toward a dimensional, severity-based approach.
Previous Ways of Recognizing Anxious Distress
The term anxious depression has long been used to describe the emergence of anxiety in the setting of a depressive episode, and synthesis of research findings has been complicated by heterogeneous definitions of anxious depression. Diagnosis in research settings has typically been based on a DSM or ICD diagnosis of MDD presenting with subthreshold anxiety symptoms measured using a variety of tools and score cutoffs,6 such as the Hamilton Depression Rating Scale (HDRS) anxiety/somatization factor (score ≥ 7), the Hamilton Anxiety Rating Scale (HARS) total score, or a mix of items from scales such as the Montgomery-Asberg Depression Rating Scale (MADRS) and the Inventory for Depressive Symptomatology.7 Dr Manning said that he does not perceive a meaningful difference between previous conceptions of “anxious depression” and what is captured by the anxious distress specifier—therefore, as was true previously, best practices in the clinical setting include effective screening and tracking of anxiety symptoms in depressed patients. Dr Thase felt that the specifier represents a positive change in the nomenclature in that it denotes patients who tend to have poorer outcomes. The rating scale included within the specifier allows clinicians to codify the extent of the patient’s anxiety as opposed to simply noting the presence of anxiety.
Validation
Two validation studies of the anxious distress specifier have been conducted.1,8 Using data from NESDA, Gaspersz et al1 showed that the specifier better predicted chronicity, time to remission, and functional disability outcomes in MDD patients than did DSM-IV anxiety disorder diagnoses.1 Zimmerman et al8 modified the Clinically Useful Depression Outcome Scale to include items keyed to the specifier and found their scale to be a reliable and valid predictor of anxiety symptoms; further, the specifier was associated with greater functional impairment and poorer quality of life.
Because such a large proportion of patients meet specifier criteria, Dr Manning thought that denoting the absence of anxious distress might have been more appropriate, or that the DSM-5 requirement of meeting 2 of 5 criteria for the majority of the depressive episode could be raised to 3 of 5 criteria to more clearly differentiate severe distress. Along these lines, Dr Trivedi noted that the number of anxious symptoms exhibited by the patient is significant—those with 3 or 4 symptoms may be at greater risk and have poorer outcomes than those with 1 or 2. He speculated that although the specifier’s minimum cutoff of 2 symptoms might not have been derived from hard data, it had most likely been chosen because it made good sense to the experts formulating the criteria.
- Between 40% and 60% of patients with major depressive disorder (MDD) also have anxiety symptoms. An anxious distress specifier was included in DSM-5 to help identify and treat patients with this symptom profile.
- Anxious distress in MDD is associated with greater depression severity and poorer response to treatment, as well as lower functioning and increased suicidality.
- Objectively measuring symptoms at each visit helps clinicians diagnose more precisely and make better-informed treatment choices. Several brief, easy-to-administer scales are available for this purpose.
- SSRIs and psychotherapy are first-line treatment options. If these yield inadequate response after a sufficient length of time, switching antidepressants or combining treatment modalities, such as augmenting with atypical antipsychotics, should be considered.
Time Course
Anxious symptoms often precede depressive symptoms in patients who develop both, as noted by Drs Trivedi and Thase. In the WHO World Mental Health Surveys, 68.0% of those with lifetime anxious MDD reported onset of anxiety disorders at earlier ages than MDD.9 A temporal sequencing analysis10 of NESDA data found that anxious symptoms emerged before depressive symptoms in about 57% of individuals with both; depressive symptoms were first in only 18% of cases, and in 25% of cases, they emerged simultaneously. An onset pattern with anxiety first was associated with longer symptom duration, earlier age at onset of the first disorder, and a greater number of fear symptoms.10
Patient Characteristics
Anxious depression has been shown to be significantly more common among women11,12; African Americans and Hispanics13; and those who are married, divorced, or widowed versus single.13 Associations with lower education level,13-15 unemployment,12 and being on public insurance13 have also been demonstrated. A NESDA analysis10 found, further, that those with anxious depression had higher neuroticism scores and lower levels of extraversion, agreeableness, and conscientiousness. An association with childhood trauma was mentioned in the meeting; supporting that point, Hovens and colleagues showed greater risk of onset16 and a more chronic course of depression and comorbid anxiety/depression17 in those with childhood emotional neglect and psychological abuse. Earlier age at onset of MDD has also been reported; Fava et al12 found that the mean age at onset of first major depressive episode for those with anxious versus nonanxious depression was 24.7 vs 25.5 years (P = .0064).
Response to Treatment and Remission Rates
The participants agreed that anxious patients are known to be more difficult to treat, more severely depressed, and less responsive to treatment. Depressed patients with anxiety take longer to respond to medications—in Dr Thase’s estimation, 2 to 4 weeks longer. They also have a longer time to remission (Figure 1)15 and are less likely to experience remission.18 Further, anxiety symptoms tend to linger: Romera et al19 showed that after 3 months of antidepressant treatment, 78.2% of patients had residual anxiety symptoms. Remission rates are significantly lower in patients with anxious depression following initial antidepressant treatment. In a NESDA-based sample of 149 patients with MDD,14 those who met anxious distress specifier criteria were significantly less likely to achieve remission after 2 years of adequate antidepressant treatment: 43.2% still had MDD, versus only 21.3% of those without the specifier. The specifier also predicted more severe depression at both 1-year and 2-year follow-up. The specifier was better at predicting lower remission rates than were comorbid DSM-IV anxiety disorders. Findings have not been completely uniform, with one large study11 showing no difference in time to response at 12 and 28 weeks between MDD patients with and without anxious features.
CONSEQUENCES OF LACK OF DIAGNOSIS AND TREATMENT
Effects on the Patient’s Family and Children
Symptoms of depression and anxiety have consequences not just for the affected individual but also for the person’s family. Drs Thase and Manning found the Sequenced Treatment Alternatives to Relieve Depression (STAR*D)-Child study,20 which looked at the impact of mothers’ unremitted depression on their children, particularly meaningful. Effective treatment of women was associated with reduction of symptoms and diagnosis rates in their children in essentially a dose-response relationship—and more persistent depression in the mothers was linked to development of more psychiatric difficulties in their children. Dr Thase commented astutely that people who might be ambivalent about pursuing treatment for depression need look no further than the well-being of their children, because it is directly dependent on their own well-being.
Poorer Prognosis
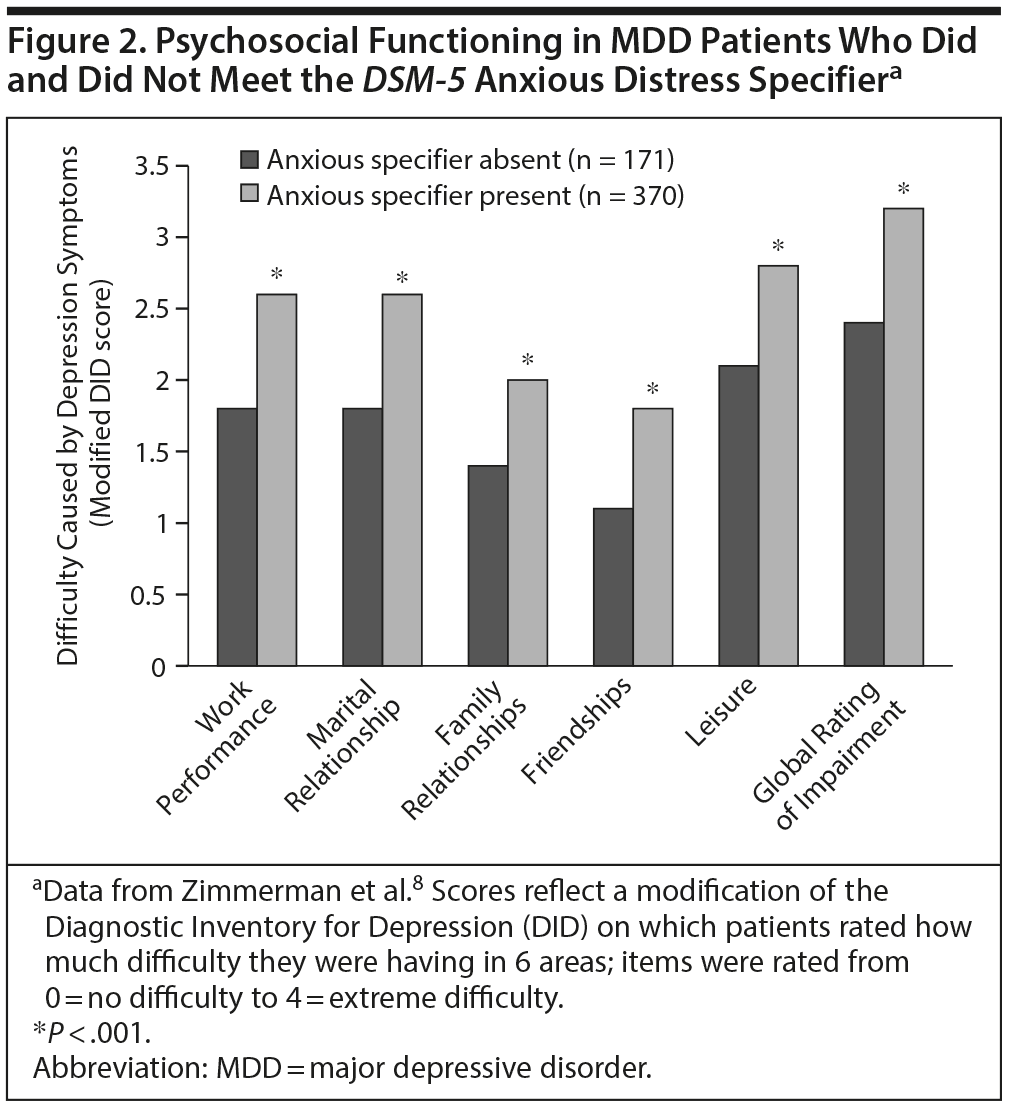
Functioning. The participants concurred that in depressed patients, anxiety is a signaler of greater suffering, mental anguish, and lower functioning. Among psychiatric outpatients, Zimmerman et al8 found significantly lower psychosocial functioning, reduced life satisfaction, and poorer quality of life among depressed patients who met the DSM-5 anxious distress specifier (Figure 2). Worse cognitive function and poorer work and social adjustment have also been demonstrated.11 A primary care study21 showed that about 10% of eligible patients screened in clinic waiting rooms reported unrecognized and untreated anxiety symptoms. These patients reported significantly worse functioning on both physical and emotional measures than nonanxious patients. A striking finding was that the anxious patients reported reduced functioning within ranges to be expected in chronic physical diseases such as diabetes and congestive heart failure. The most severe reductions were reported when anxiety was mixed with depressive disorders.
Dr Weisler pointed to the cascading effects of poor functioning. Poorer functioning can lead to dropping out of high school or college, which leads to increased mortality rates. In fact, mortality attributable to low education is comparable to that attributable to smoking.22
Sleep problems. Sleep problems were cited as being particularly troublesome in anxious depression. For example, Fava et al12 demonstrated substantially higher insomnia rates in anxious depression (82%, 90%, and 66% for early, middle, and late insomnia) versus nonanxious depression (60%, 77%, and 47%). Dr Trivedi noted further that there seems to be a dose-response relationship between anxiety severity and sleep problems, in that patients who have 3 to 5 of the symptoms in the anxious distress specifier seem to have higher rates of sleep disorders than those with 2 or fewer. The National Comorbidity Survey Replication linked anxiety to comorbid sleep disorders, finding a prevalence of 37% among those with any anxiety disorder (among those with any mood disorder, the prevalence was 25%).23 Sleep complaints in those with anxious depression are especially troubling because of the link between sleep problems and suicidality.24
Suicidality. The American Psychiatric Association (APA) practice guideline for MDD lists severe anxiety as a factor to consider when assessing suicide risk and recommends treating it as a modifiable risk factor to lower the risk of suicide in addition to treating the depressive episode.25 Anxious distress in those with MDD has been associated with increased suicidal ideation13,14 and attempts.13 Dr Weisler mentioned that as the number of anxiety symptoms increases, so does suicidality. Impulsivity was also identified by Drs Weisler and Thase as a key factor: according to Dr Thase, attempts among those with anxious depression “are not the planned suicides; not the ones in which the person has written a note, but rather the ones in which the person shoots themselves or jumps off the roof because they can’ t stand feeling the way they do any longer.” Further, he believes that an intolerance of strong negative affects plays a part in the development of suicidality in these individuals and that the intensity of the anxiety is the “coup de gr×¢ce.” Impulsivity makes suicidality in these patients more difficult to predict and prevent.
Increased cardiovascular risk. The American Heart Association classifies MDD as a tier II moderate-risk condition associated with accelerated atherosclerosis and early development of cardiovascular disease,26 and a meta-analysis27 showed an overall odds ratio (OR) of 1.60 for the onset of myocardial infarction in those with depression. The cardiovascular risk associated with anxious distress may be especially high. Almas et al,28 in a longitudinal cohort study, found that DSM-5-defined anxious distress was associated with a cardiovascular disease risk with an OR of 2.1, higher than the OR associated with depression (OR = 1.9).
CASE VIGNETTE 1*
Shari, a 36-year-old woman, presented with psychiatric symptoms that had increased over the past 6 months, beginning when her father had a myocardial infarction. Although he recovered, Shari’s distress did not, and her worries shifted to matters such as her children’s well-being, her husband’s heart attack risk, and the possibility of unlikely events, such as auto accidents. Shari’s predominant mood was low, but she was often anxious and tense and reported increased musculoskeletal pain. She felt guilty for letting her family down, reported poor concentration, and could not fall asleep for 1-2 hours each night despite being tired. Her appetite was decreased except for occasional episodes of “nervous” binge eating. She was less interested in spending time with her friends, and she had diminished enthusiasm for sex. She did not have suicidal ideation but sometimes wondered if she could go on feeling this way. She also said that the current anxious and depressive symptoms were unusual for her.
Case Practice Question 1
Which aspect of Shari’s presentation is not an indicator of anxious distress?
- Poor concentration
- Low mood
- Worries about possible future events
- Tension
Discussion of Case Practice Question
Preferred response: b.
Explanation: Shari’s symptoms are consistent with a diagnosis of major depressive disorder with anxious distress. While low mood is a symptom of major depressive disorder, it is not an anxious distress specifier criterion.
Shari’s symptoms, which had continued for a significant period of time, are consistent with a DSM-5 diagnosis of MDD with anxious distress. Her diminished concentration, low mood, and sleep disturbance all point to an MDD diagnosis; the additional presence of tension and excessive worry and her persistent feeling that something awful could happen at any time further suggest that application of the anxious distress specifier is appropriate.
SCREENING AND MEASUREMENT
Measurement-Based Care
The participants agreed that it is essential that clinicians screen for and track anxiety and depression symptoms, and the APA recommends measuring outcomes as part of good clinical practice.25 Measurement-based care can be summarized as systematic and quantitative measurement of symptoms at each visit; other parameters such as treatment adherence, psychosocial functioning, and side effects should also be assessed.
Measurement-based care is especially important in patients who have depression with co-occurring anxiety symptoms because, as Dr Manning put it, “People will say, ‘ My nerves are bad,’ and they typically talk about that in relationship to anxiety or irritability. That’s one reason we measure with scales: to get discrimination around the symptoms they’ re having so that we can make a precise diagnosis and prescribe rational treatment.” He also underscored the importance of asking questions about the patient’s own goals for treatment and approaching the patient empathetically: “There’s the surface analysis, and then there’s the collaborative, empathetic kind of conversation: ‘ So, how can I best help you? What were your hopes for being here today? And I know that everybody who’s depressed thinks about death, so what you have been thinking about death?’ I’ m more likely to ask that question and expect more serious answers if the patient is severely anxious.” Further, he explained, measurement is a part of effective clinical care regardless of whether the symptoms are physical or mental:
I would never treat a patient’s hypertension without measuring their blood pressure. And, of course, I would never get just a “clinical global impression” of their high blood pressure. If I’ m treating diabetes, I’ m going to measure glycosylated hemoglobin and ask to see self-monitored blood glucose data. In the last 15 years, measurement has been the best help for me in improving how I treat patients.
According to Dr Trivedi, the value of measurement tools lies in the fact that they provide strong evidence regarding symptoms, function, quality of life, and side effects so that the subsequent patient interview can be more focused, which can improve quality of care and increase remission rates. Dr Thase noted that the most significant legacy of STAR*D was perhaps its implementation of measurement-based care: “Clinicians do pay attention to feedback that patients aren’ t getting better or are suffering from side effects. But if clinicians aren’ t asking the questions, or if patients don’ t have the opportunity to let them know that things aren’ t going well, then the feedback doesn’ t occur.” He also hoped that the APA practice guideline update will emphasize the importance of measurement-based care in light of new evidence from randomized controlled trials (RCTs).
The shortage of primary care providers and the limited time available for evaluation were cited as contributors to underrecognition of anxiety, and Dr Weisler observed that symptoms are often not elicited or measured until the patient is referred for psychiatric care. To assess at an earlier stage whether a patient meets the anxious distress specifier criteria, a brief and reliable measurement is needed, and Dr Manning felt that if a well-validated discriminatory scale for measuring anxious distress were available, primary care providers would quickly adopt it.
Screening Tools for Clinical Settings
Several easy-to-administer, well-validated, and sensitive scales are available for measuring depressive and anxious symptoms (Table 1).
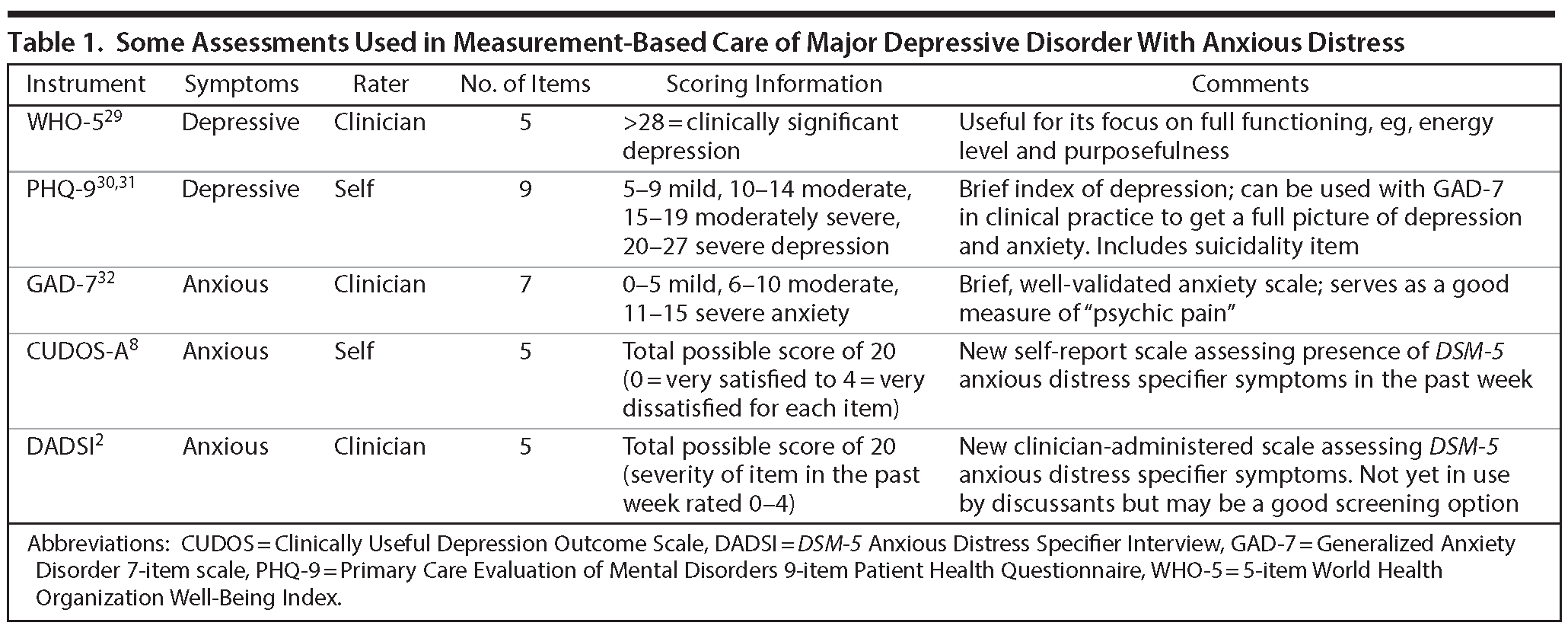
WHO-5. The 5-item World Health Organization Well-Being Index (WHO-5) is a short rating scale measuring subjective well-being. The items consist of 5 positively phrased statements (eg, “I have felt cheerful and in good spirits”) and are rated on a 6-point Likert scale based on the proportion of time, over the last 2 weeks, when the respondent would agree with the statement. The WHO-5 has good validity as a depression screening measure.29 Drs Trivedi and Manning agreed that this scale was a meaningful assessment in clinical practice, particularly because of its focus on elements important to patient functioning such as energy, restfulness, and purposefulness.
PHQ-9. The Primary Care Evaluation of Mental Disorders 9-item Patient Health Questionnaire (PHQ-9)30 is a self-rated depression measure that focuses exclusively on DSM-based symptoms.30 The frequency of 9 symptoms is rated on a scale of 0 to 3: not at all, several days, more than half the days, and nearly every day, respectively, in the last 2 weeks. A follow-up question inquires about the patient’s functioning. Katzelnick et al31 demonstrated that the PHQ-9 allowed clinicians to successfully determine rates of response (defined as a score < 10) and remission of depressive symptoms (score < 5) at 12 and 24 weeks. Dr Manning mentioned that one advantage of the PHQ-9 is that it allows for ongoing inquiry about thoughts of death or distress.
GAD-7. In clinical practice, the well-validated Generalized Anxiety Disorder 7-item scale (GAD-7)32 is often used. Dr Manning mentioned that it has a large evidence base and is the screener used in his practice. The participants praised it for being effective and specific. Each item represents an anxiety symptom, and patients indicate how bothered they have been by the symptom over the last 2 weeks on a scale of 0 to 3. Its advantages include its short length and self-administered format. Dr Trivedi noted that the PHQ-9 and GAD-7 can be combined with a cognition measure to comprise a full clinical picture of symptoms.
CUDOS-A. To devise a scale keyed specifically to the DSM-5 anxious distress specifier, Zimmerman et al modified the Clinically Useful Depression Outcome Scale to include items capturing the anxious distress specifier (CUDOS-A).8 This self-report scale consists of 5 items that correspond to the 5 criteria in the anxious distress specifier, and each is rated on a 5-point ordinal scale. Zimmerman et al demonstrated good internal consistency, reliability, and validity,8 and advantages include its brevity and self-administered format. While the specifier requires anxiety symptoms to be present for the majority of days during an episode, the CUDOS-A measures symptoms cross-sectionally, capturing only those present that week.
DADSI. Zimmerman and colleagues2 also developed the DSM-5 Anxious Distress Specifier Interview (DADSI), a clinician-rated scale assessing the 5 symptoms in the specifier. This scale inquires about the severity of symptoms over the past week as well as whether the symptoms have been present for the majority of the episode. In a sample of 173 depressed patients from the Rhode Island Methods to Improve Diagnostic Assessment and Services project, 78% met criteria for the anxious distress specifier.2 The authors demonstrated reliability and validity and found that the scale was sensitive to detecting improvement. Dr Manning commented that the DADSI is still a new scale and is unproven in broad practice in comparison to an instrument such as the GAD-7. Although the participants do not currently use the CUDOS-A or DADSI, they did praise Zimmerman’s approach, and Dr Weisler mentioned that the scale is quick to administer.
Electronic Assessment
Dr Weisler mentioned an ongoing study with which he is involved in which patients rate symptoms using their phones via an app; not only is this convenient, but it allows clinicians to more accurately and effectively track changes in depression or anxiety symptoms. Dr Manning commented that, ideally, information on depressive and anxious symptomatology would be digitized and collected via a kiosk or a smartphone app that would interface with the electronic health record to minimize workload.
Further, Dr Trivedi relayed that his group has developed software called VitalSign6 to capture PHQ-9 and GAD-7 data.33,34 Designed for use in primary care, pediatric, and specialty clinics, the software collects data on symptoms, side effects, and adherence. Patients complete the measures on an iPad. Dr Trivedi commented, “There is an indirect but a very solid benefit from these routinized itemized measurements—patients become much more educated about what the goals are, what the targets for their treatments are, and what they are looking for in terms of improvement or lack thereof. You can then target patients’ treatments to those goals, and this is closer to shared decision making.” Dr Trivedi also relayed an insight from patient advisory focus groups: for some patients, self-rated symptom and side effect assessments may be more effective than clinician interview in eliciting symptoms. “They feel pressured when a doctor is asking, ‘ Well, how’s your sleep? How’s your appetite?’ With a self-rated measure, they are more relaxed and have more time available to answer. Patients appreciate that.”
CASE VIGNETTE 2
Charlie, a 25-year-old man, presented in a primary care clinic to obtain herpes prophylaxis. He was getting married soon and also requested medication to help manage anxiety in relation to the wedding. Charlie had recently returned from 2 tours of duty in the Middle East, and though he had tried to make an appointment in the VA system to address his anxious symptoms, he had encountered great difficulty.
Case Practice Question 2
What is an example of a brief scale the clinician could administer to get a better picture of Charlie’s overall mental well-being?
- Hamilton Depression Rating Scale
- Primary Care Evaluation of Mental Disorders 9-item Patient Health Questionnaire
- World Health Organization Well-Being Index
- Comprehensive Psychopathological Rating Scale
Discussion of Case Practice Question
Preferred response: c.
Explanation: The World Health Organization Well-Being Index (WHO-5) measures subjective well-being and can be administered quickly in a clinical setting.
The clinician administered the GAD-7, PHQ-9, and WHO-5 to get a complete picture of Charlie’s anxious and depressive symptoms and level of functioning. Charlie reported excessive worry and anxious symptoms as the primary cause of his mental distress, but the assessments also indicated significant depressive symptoms, such as low mood and decreased energy levels, that had emerged subsequent to the anxiety and required attention.
Charlie’s life situation should prompt a number of issues for the clinician to consider: How will his anxiety and mood symptoms affect his new marriage? Will his wife be at greater risk of suffering interpersonal violence? If he has children, will the quality of his parenting be affected, and will his children be more likely to develop mood or anxious symptoms, as has been shown for symptomatology in mothers?20 These questions illuminate the possible consequences of not detecting and treating depression and anxiety. If the primary care clinician had not used a measurement-based care approach, employing 3 structured assessments, the patient’s significant depressive symptoms may have gone unaddressed entirely.
TREATMENT STRATEGIES
The participants emphasized that even if an antidepressant is efficacious for a patient’s depressive symptoms, his or her anxiety may not improve. The participants also recognized the importance of individual patient characteristics and symptom profiles. Patients, as well as their family members, may have received multiple agents before, so the history of response should be considered. Dr Weisler mentioned that pharmacogenomic testing may sometimes be of value in determining which antidepressant and/or dose to use in ultrarapid or poor metabolizers (as well as whether l-methylfolate augmentation might be helpful, for example, in patients with T or TT MTHFR alleles). Anxiety subtype also matters: Dr Weisler commented that anxiety symptoms related to panic or obsessive-compulsive disorder or posttraumatic stress disorder (PTSD) might lead him to choose a drug that is FDA approved or studied for treating those disorders. In the case of PTSD symptoms, he might avoid a benzodiazepine, since lack of efficacy and problems related to learning have been demonstrated in PTSD for that medication class.35 He also advised taking a few minutes to assess for bipolarity by inquiring about personal or family history of mania/hypomania, earlier onset, and higher number of lifetime mood episodes and employing a simple tool such as the Mood Disorder Questionnaire. Dr Weisler stated that his strategy for pharmacologic treatment of MDD with anxiety would be this:
- Step 1: Prescribe an SSRI or SNRI that has FDA approval both for anxiety and for MDD.
- Step 2: Optimize the dose and duration of this treatment.
If partial or nonresponse occurs’ ¦
- Step 3: Add buspirone up to 60 mg/d (but not bupropion, due to increased initial anxiety).
If partial or nonresponse occurs’ ¦
- Step 4: Depending on the patient’s medical history and anxiety severity, add a second-generation antipsychotic that has FDA approval for MDD.
Dr Manning commented that if a major guideline (eg, APA, CANMAT) were to recommend proven and efficacious interventions for these patients, he would follow them. The 2016 CANMAT guidelines36 recommend choosing an antidepressant with efficacy in generalized anxiety disorder for patients with anxious distress, but this recommendation is level 4 (expert opinion) evidence; the 2012 guidelines specific to MDD with comorbid anxiety37 found no strong basis for altering pharmacotherapeutic treatment in the presence of anxiety. The 2009 APA guidelines25 predate the anxious distress specifier. They do not substantially differentiate initial treatment choice for patients with anxiety symptoms but do mention a role for augmentation with benzodiazepines or sedative hypnotics; neither is a level 1 (substantial clinical evidence) recommendation.
Special Considerations
Dosing. The APA and CANMAT practice guidelines recommend lower starting doses with slow upward titration for patients with significant anxiety.25,37(p14)
Tolerability/side effects. Asked which side effects might be particularly prevalent in patients with MDD and anxious distress, Dr Thase replied, “All of them.” He explained further: “Think of the predisposition to anxiety as an amplifier in the limbic system of preoccupation and concern with distress. Any somatic cue is a potential danger signal. In studies of depression—with all types of antidepressants—the anxious depressed patients nearly always have higher levels of a broad array of autonomic, gastrointestinal, and CNS side effects.” Dr Weisler stressed the importance of weighing benefits and drawbacks. In certain patients, oversedation may be an issue; in others, extrapyramidal symptoms may complicate the picture. Gradual dose increases and close monitoring of progress are key.
Chan et al,11 in the large Combining Medications to Enhance Depression Outcomes (CO-MED) study, found higher side effect burden in depressed patients with anxiety symptoms (P = .0009) even if they were not receiving higher doses. Gaspersz and colleagues14 found that patients with the DSM-5 anxious distress specifier had a higher number of side effects; 41% of those with the specifier had at least 4 side effects, versus only 20% of those without the specifier. (Logistic regression revealed no effect of the specifier on reporting of 1 to 3 side effects.) A distinct neurobiological profile in these patients (as demonstrated by Ionescu et al38) may be responsible for differences in side effect prevalence.
Adherence issues. The importance of ensuring medication adherence was discussed; Dr Weisler remarked, “Adhering to treatment is one of the biggest problems that our patients have. One of the advantages I’ ve found in looking at a controlled drug registry is that, in addition to seeing other potential providers and drugs with abuse potential, I can find out what pharmacies patients are using. I can then call their pharmacy and see if the patient is filling other medical and psychiatric prescriptions. Anxiety can drive nonadherence, partly because if a person is anxious and worried, they tend to be more forgetful.”
Antidepressants
SSRIs. Selective serotonin reuptake inhibitors (SSRIs) are a mainstay of first-line pharmacologic treatment across different clinical guidelines for depression.25,36 For patients with anxious depression, though, SSRI monotherapy may prove inadequate; Dr Manning noted that in many patients they are only modestly helpful for anxiety. In level 1 of STAR*D, only 22% of patients with anxious depression reached remission with 12 to 14 weeks of citalopram monotherapy.13 Fava and coworkers39 showed higher remission rates but no significant differences in efficacy or tolerability among fluoxetine, sertraline, or paroxetine for acute treatment of MDD in patients with anxious depression. Thaler and colleagues’ 40 review of the literature also indicated a lack of difference among second-generation antidepressants for these patients.
SNRIs. If monotherapy does not bring symptom relief, tapering the medication and simultaneously titrating up an antidepressant of a different class is a possible next step. Dr Weisler felt that there is not much difference in efficacy between the SSRIs and serotonin-norepinephrine reuptake inhibitors (SNRIs; venlafaxine, duloxetine) in treating anxious depression. A post hoc analysis found greater remission rates at 6 weeks with venlafaxine versus fluoxetine41; however, a comparison of venlafaxine and sertraline showed similar symptomatic improvement.42
Bupropion. The recent large, multicenter Veterans Affairs Augmentation and Switching Treatments for Improving Depression Outcomes (VAST-D) trial43 showed a greater incidence of anxiety with bupropion versus atypical antipsychotic augmentation. Trivedi and colleagues,44 however, showed comparable effectiveness of bupropion and sertraline in depressed patients with anxious symptoms, and the CANMAT guidelines36(p545) state no difference in efficacy among SSRIs, SNRIs, and bupropion for anxious distress.
Antidepressant combination. No antidepressant is approved by the US Food and Drug Administration (FDA) as combination therapy for treatment-resistant MDD; however, clinicians often combine antidepressants as a second-step approach. The CO-MED study11,45 evaluated first-step combinations of escitalopram + placebo, escitalopram + sustained-release bupropion, and extended-release mirtazapine + venlafaxine for both acute (12-week) and continuation (28-week) therapy. Across the treatment groups, at 28 weeks, 44% of those with and 45% of those without anxious features achieved remission. The authors found no association between presence of anxious features at baseline and differential treatment outcomes among the treatment groups.11 CO-MED found no advantage for antidepressant combination as first-line therapy (bupropion + escitalopram or mirtazapine + levofloxacin) versus escitalopram alone,45 only a greater side effect burden. Dr Thase commented, though, that combinations might be useful for certain subsets of patients, such as those with greater severity of symptoms or treatment resistance.
Nonpharmacologic Strategies
CBT. Cognitive-behavioral therapy (CBT) may be used as a first-line treatment for MDD,25(p17) particularly in patients who express a preference for it. It may be used instead of or in addition to pharmacotherapy. At level 2, STAR*D found no significant differences in response and remission outcomes with CBT, as either monotherapy or adjunctive treatment, versus pharmacotherapy alone. Time to remission was greater with CBT augmentation than with medication augmentation (55 vs 40 days) but did not differ for switching to CBT versus switching to medication.46 A subanalysis comparing patients with anxious and nonanxious depression47 showed no significant interaction between anxious depression and treatment assignment, but significantly lower response rates were seen for anxious depression versus nonanxious depression in the CBT switch group (14% vs 36%), and lower remission rates were seen in both the switch (21% vs 51%) and augmentation (14% vs 38%) groups.
Two or 3 hours of therapist time spread over 6 weeks was recommended by Dr Thase for achieving optimal results, and he commented that therapist engagement is important even if therapy is delivered electronically: “Depression undermines the ability to learn new material, focus on it, and follow through with the practice. Evidence-based psychotherapy can be learned online, but without the therapist’s guidance and encouragement, the book often goes ‘ unopened,’ so to speak.” Dr Trivedi relayed that as part of the VitalSign6 quality improvement project, his group recently completed a study34 evaluating the outcomes of 8 sessions of behavioral activation delivered through teletherapy. The sample consisted of 74 low-income primary care patients with moderate depressive and anxious symptoms. The majority of patients who received at least 1 therapy session achieved depression remission, and patients completing at least 4 sessions demonstrated substantial decreases in depression (median PHQ-9 score decrease of 10.0) and anxiety (median GAD-7 score decrease of 8.0) symptoms at the final session.
Physical exercise. The participants agreed that physical exercise can be an effective adjunct, and Dr Weisler pointed to Dr Trivedi’s contributions in this area. In a sample of sedentary individuals with SSRI-resistant MDD, Trivedi and colleagues48 compared two 12-week exercise interventions with energy expenditures equivalent to walking 210 minutes per week at 4 miles per hour or 75 minutes per week at 3 miles per hour. Covariate-adjusted remission rates were 15.5% for lower-dose and 28.3% for higher-dose exercise augmentation, leading to an NNT of 7.8. Trivedi et al48 point out that remission rates with exercise are comparable to those seen with an SSRI/bupropion combination and that their results are within the range of findings that warrant change in clinical practice. According to Dr Trivedi, for a person weighing about 80 or 90 kg, about 1,200-1,400 calories of energy expenditure per week is needed to see an effect. This translates to 40-45 minutes of moderate to vigorous exercise 3 times per week. The rigor of the exercise should be significant, and the patient should not be able to have a conversation when exercising. Heart rate should be about 80% of the maximum rate for the patient’s age.
Relaxation exercises and good sleep hygiene. Echoing earlier points about sleep, Dr Weisler said that he has found sleep hygiene recommendations, as well as relaxation exercises, to be useful for many patients. He noted that resources in these areas are freely accessible to patients from a number of online providers and that they can make a substantial difference. He also mentioned the value of assessing for and treating sleep apnea because of its link with depression.49
Augmentation Strategies
It was noted that the poorer prognosis associated with anxiety symptoms demands a greater need to combine different treatment modalities. If a patient does not respond to initial treatment of optimal dose and duration, or if residual symptoms are present, augmentation strategies involving a medication targeting another neural circuit can be considered.
Atypical antipsychotics. At present, 3 atypical antipsychotics are FDA-approved for adjunctive use in treatment-resistant MDD: aripiprazole, brexpiprazole, and extended release quetiapine (quetiapine XR). A fourth agent, olanzapine, is approved for use in treatment-resistant MDD in combination with fluoxetine. Meta-analyses have demonstrated greater efficacy of these medications versus placebo as augmentation in patients with treatment-resistant MDD. Nelson and Papakostas50 found ORs of 1.69 and 2.00 for response and remission, respectively; Spielmans et al51 also showed statistically significant effects on response and remission rates as well as clinician-rated depression severity measures.
Aripiprazole. Trivedi et al52 demonstrated efficacy of adjunctive aripiprazole in a pooled analysis of two 14-week studies that included patients who had shown nonresponse to 1 to 3 previous antidepressant trials. Both the anxious and nonanxious depression groups showed greater improvement in MADRS total score with aripiprazole versus placebo (decreases of 8.72 and 8.61 in the anxious and nonanxious groups, vs 6.17 and 4.97 with placebo). Additionally, the VAST-D study43 demonstrated a significantly better remission rate for augmentation with aripiprazole versus switching to a different antidepressant (relative risk = 1.3; P = .02). Akathisia has been reported as the most common adverse event with aripiprazole.53 Trivedi and colleagues52 showed that although discontinuation due to adverse events was low, and aripiprazole was generally well tolerated, it was associated with a higher rate of akathisia (24.6%) than placebo (4.4%).
Brexpiprazole. Drs Weisler and Thase pointed to a post hoc analysis of 2 RCTs7 that indicated efficacy for brexpiprazole in MDD patients who had nonresponse to antidepressant monotherapy. McIntyre et al7 defined anxious distress using proxies for DSM-5 criteria, and 55% of the sample met the criteria.The studies compared brexpiprazole at doses of 1, 2, and 3 mg and placebo as adjuncts to antidepressant treatment. Brexpiprazole showed greater improvement than placebo on MADRS score change at week 6 in patients both with and without anxious distress; in the anxious distress group, the least squares (LS) mean difference in MADRS score change was -2.95 vs placebo (P = .002) with the 2-mg dose. An RCT54 in antidepressant nonresponders yielded similar findings; the LS mean difference in MADRS score from placebo for brexpiprazole at week 8 was -2.98 in patients with anxious distress (P = .0099). Moderate weight gain and akathisia are reported with brexpiprazole.55
Quetiapine XR. Bandelow et al56 investigated efficacy of quetiapine XR at 150-mg and 300-mg doses as adjunctive therapy in a pooled analysis of two 6-week RCTs. When anxious depression was defined as a HARS score ≥ 20, significant MADRS score reductions vs placebo at week 6 were found for both doses in those with nonanxious depression, but only for the 300-mg dose in those with anxious depression. With an HDRS anxiety/somatization score ≥ 7 as the definition, MADRS score reductions were significantly greater for both doses starting at week 1 (week 6: 150 mg/d, P < .01; 300 mg, P < .001) in patients with anxious depression, but for neither dose in those with nonanxious depression. Adverse events commonly associated with quetiapine are weight gain, metabolic disturbances, and sedation.57
Olanzapine. Data specifically on olanzapine and anxious depression are lacking. In a 12-week study,58 olanzapine/fluoxetine combination (OFC) was associated with rapid antidepressant effect and improvement in depressive symptoms compared with antidepressant monotherapy in MDD patients during the first 6 weeks of treatment, but at study endpoint, the groups were not significantly different. Two other large 8-week studies compared OFC to olanzapine and fluoxetine monotherapies in nonresponders to fluoxetine monotherapy.59 One study showed no significant between-therapy difference in MADRS score change; however, the pooled analysis showed a remission rate of 27% for OFC (vs 17% and 15% for fluoxetine and olanzapine monotherapies), as well as a shorter time required for 25% of patients to achieve therapeutic responder status (30 days with OFC vs 55 and 53 days for fluoxetine and olanzapine monotherapies). Somnolence, sedation, and weight gain are commonly observed with olanzapine.60
Use in clinical practice. Dr Manning commented that he has seen an increase in prescription of second-generation antipsychotics among primary care providers for treating symptoms of anxiety as well as depression, and he felt that the published data on metabolic issues have not affected practice much. He advises primary care and mental health care providers to obtain a comprehensive metabolic panel, lipid panel, and hemoglobin A1c measurement at the start of therapy, to repeat those measurements within 3 months, and, depending on the results, to obtain them thereafter every 6 months to once per year. He explained why: “The connection between depression and cardiovascular health and the importance of treating depression in cardiovascular patients demands that we monitor for ways we might be worsening a patient’s metabolic status, and this must be balanced for each individual.” Dr Weisler pointed out that efficacy must be balanced with side effect risk, citing a meta-analysis50 of atypical antipsychotic augmentation that showed a higher remission rate for these agents versus placebo (pooled rate of 30.7% vs 17.2%; P < .00001), but also a higher rate of discontinuation due to side effects (OR = 3.91; P < .00001). Therefore, it is important to monitor the effects of treatment, including clinical improvement and adverse events, to optimize response and adherence in each individual.
Buspirone. Buspirone enhances SSRI activity through the 5-HT1A receptors61 and is FDA-approved for short-term treatment of anxiety symptoms. Physical dependence is not a concern.62 Level 2 of STAR*D compared bupropion versus buspirone as augmentation of citalopram following initial nonresponse to monotherapy and found remission rates of 36.7% and 39.2%, respectively, in nonanxious patients; however, in those with anxious depression, the rates were only 17.9% and 9.2% (Figure 3).13,63 Dr Trivedi called the results surprising, in that the investigators expected to find that buspirone would be superior in anxious depression—this was not the case, and remission rates in those with anxious symptoms were startlingly low with either agent. According to Dr Trivedi, the findings demonstrated the existence of “a subgroup of patients who have significant anxiety and meet the criteria for the anxious depression specifier and who don’ t do well on monoaminergic agents—this is true for any of the antidepressants.”
Benzodiazepines. Although benzodiazepines do not treat depressive symptoms, they are often used as augmentation for anxiety and/or sleep problems. The APA Practice Guideline25(p19) includes them as an option that “may be recommended on the basis of individual circumstances” if anxiety or insomnia is prominent. This course of action would follow first trying SSRI or SNRI monotherapy. Short-term use (ie, < 4 weeks) in combination with an antidepressant has been shown to lessen anxiety.64 However, their effectiveness must be balanced against the abuse potential. Dr Thase recommended making, at the outset, a commitment with the patient to soon taper the benzodiazepine dose and find other combination strategies. In older patients, benzodiazepines can present an elevated fall risk, but Dr Thase did not believe that they act as an accelerant of dementia—rather that unremitting anxiety is a more likely accelerant. Dr Weisler agreed, noting that for some patients, lack of sleep can be another aggravating factor for dementia. Overdose dangers were discussed, and Dr Manning cited concerns among primary care clinicians about prescribing benzodiazepines, as well as a focus on limiting exposure to the short term. Dr Weisler noted that many patients with depression and anxiety take opioids and that the combination with benzodiazepines can be dangerous, possibly leading to difficulty breathing or even death. Use of prescription drug monitoring programs and electronic health records has been suggested as a way to help clinicians identify risky use of these medications.65 Dr Weisler cited a recent report66 that the 16% of Americans who have mental health disorders receive over half of all opioids prescribed in the United States, as well as data showing that 30% of individuals dying due to opioid-related causes in 2010 also had benzodiazepines in their system.65 He emphasized that improving pain management in this population is critical to reduce opioid dependency.
Other medications. Thyroid hormone supplementation may be useful in some patients with treatment-resistant depression; evidence in the literature is mixed.67 Lithium is another augmentation option, with a meta-analysis68 confirming its effectiveness. A third option is l-methylfolate, which is well tolerated and also may be useful in some patients.69
In patients with sleep problems, nonbenzodiazepine sedative hypnotics such as eszopiclone, zaleplon, and zolpidem may be helpful as augmentation and present less potential for dependence than benzodiazepines. Fava et al70 showed significant reductions in total HDRS-17 score and insomnia severity, though not in HDRS anxiety/somatization score, in MDD patients with anxious symptoms receiving eszopiclone with an SSRI.
CASE VIGNETTE 3
Lucille, a 62-year-old woman, presented with a major depressive episode and had long struggled with recurrent depression. The current episode had begun 4 weeks before with no clear precipitants, although relationship stress had been associated with prior recurrences. Her youngest child had recently left for college. Lucille had a markedly depressed mood and had withdrawn from many activities, though she continued going to work. Her concentration was so poor that she struggled even to read the newspaper, and her energy was low. Her sleep was disrupted, with initial insomnia of 2-3 hours, frequent awakenings, and total sleep time averaging 5 hours. She noted decreased appetite and felt persistent nervous tension and restlessness. Lucille sometimes fretted that she had not been a good enough mother, but she also noted that these feelings were excessive. Panic-like feelings occasionally occurred, with no full panic attacks, and she at times experienced rapid heart rate and shortness of breath, especially when thinking about her children. Lucille felt a deep pessimism about her future but denied suicidal ideation. Scales were used to quantify her symptoms; she scored a 31 on the MADRS and a 24 on the HARS, and her Clinician Global Assessment was “markedly ill.”
Case Practice Question 3
Which of the following would not be considered a possible first-line monotherapy for Lucille?
- Antidepressant
- Benzodiazepine
- Cognitive-behavioral therapy
Discussion of Case Practice Question
Preferred response: b.
Explanation: Although the APA Guidelines include benzodiazepines as an augmentation option for addressing sleep and anxiety symptoms, they would not address Lucille’s depressive symptoms and therefore would be insufficient as monotherapy.
Because previous venlafaxine treatment had been unsuccessful for Lucille, sertraline was chosen and titrated to 200 mg, then decreased to 150 mg due to lethargy. After some initial improvement, at the second month her PHQ-9 score was 21, and her GAD-7 score was 14. A Clinical Global Assessment indicated she was still “moderately ill.” The physician decided to augment with brexpiprazole, beginning at 0.5 mg and titrated to 2 mg over 2 weeks. The decision to augment rather than increase the sertraline dose was related to gastrointestinal and libido-related side effects, as well as a previous SNRI failure. After 3 weeks with augmentation, her MADRS score dropped to 12, and her HARS score was 7. By week 14 (6 weeks with augmentation), Lucille was very much improved. Her Clinician Global Assessment was rated as “normal”; her self-rated PHQ-9 score was 3, and her GAD-7 score was 2, also well within the normal range. She tolerated the medications well, with only brief, mild restlessness that began when the brexpiprazole dose was raised to 2 mg and then resolved over the next week. Because Lucille had experienced multiple previous episodes, she remained on combination therapy while being monitored for adverse events.
ADOPTION OF THE SPECIFIER IN CLINICAL SETTINGS
The participants felt that the anxious distress specifier has yet to be fully adopted in clinical settings. Dr Thase commented that physicians have long been frustrated by the fact that they were making multiple diagnoses in a single patient with this symptom profile, and the specifier can serve as a way for a clinician to describe whether a patient’s presentation of depressive disorder includes more or less anxiety.
From the primary care perspective, Dr Manning said that although physicians are not yet generally aware of the specifier, it describes a common patient profile long seen in clinical practice. He doesn’ t differentiate between it and the prior concept of “anxious depression.” He also observed that the specifier has not yet gained enough momentum to be included in ICD-10. (ICD-10‘s most similar diagnosis, “mixed anxiety and depressive disorder,” requires the presence of mild symptoms of both anxiety and depression that do not meet the severity threshold for either disorder.71)
ASSESSING RESPONSE AND REMISSION
Outcomes in MDD can be categorized in 5 ways: response, remission, recovery (sustained remission), relapse (emergence of an episode during remission), and recurrence (emergence of an episode during recovery). Achieving remission is the main goal of MDD treatment.72 Measurement-based care for pursuing remission involves regular use of tools such as those described in the Screening and Measurement section. Their value lies partly in the existence of a large body of literature identifying the target scores that indicate remission. Information gathered on an ongoing basis can then be used at critical decision points to make treatment decisions in line with established algorithms and guidelines.25,73,74
Worry, poor concentration, and insomnia are all common residual symptoms, and Dr Trivedi observed that recognizing residual anxiety can be difficult because patients may say things such as simply, “I’ m not back to my normal self,” as opposed to characterizing their symptoms as anxiety-related. If the clinician fails to measure symptoms, they are easily missed. Patients should be assessed for response every 2 weeks for the first 6 weeks, and visits should be scheduled every 3 weeks thereafter until remission is achieved or treatment strategy is changed. When the patient reaches remission, assessments should continue every 3 months.72
With regard to the pattern of symptom amelioration, Dr Thase felt that there was no highly characteristic sequence in which symptoms fall away, although a need for research in this area was also noted. Further, Dr Trivedi commented that improvements in both types of symptoms are seen at generally similar rates, although anxiety may lead to slower overall response. He cited research showing that within the first 2 weeks of SSRI treatment, about 50% of patients with anxiety and depression reported improvement in anxiety; however, another 15% reported emergence or worsening of anxiety.75 In this latter subset of patients, SSRIs may be exerting an activating effect; if depression had previously been “dampening” the anxiety symptoms, they may seem to worsen as the depression improves.75
Finally, it was noted that depressive disorders are more episodic in nature, while anxiety tends to be more chronic; as a result, depressive episodes typically have clearer full remission, while anxiety symptoms, even if reduced and sporadic, often remain.
FUTURE DIRECTIONS
With regard to research efforts, Dr Trivedi emphasized a need to match more aggressive or complicated treatments for anxious distress with those patients who require them. He summarized the research gap as follows: “Treatment needs to be targeted to specific populations, and the anxiety specifier group may be one that needs more aggressive treatment.” He also thought that efforts toward adapting remission criteria to include improvement in anxious distress would be useful.
The participants expressed enthusiasm about the potential for future research on MDD with anxious distress and called for studies that, by virtue of their design, would focus purely on the anxious distress specifier (rather than measuring a “pseudospecific” effect on anxiety); for example, randomization could be stratified based on the presence of the specifier. Such efforts could then help the field move toward discovering the optimal treatments to provide relief for these patients.
Published online: November 28, 2017.
Find more articles on this and other psychiatry and CNS topics:
The Journal of Clinical Psychiatry
The Primary Care Companion for CNS Disorders
REFERENCES
1. Gaspersz R, Lamers F, Kent JM, et al. Longitudinal predictive validity of the DSM-5 anxious distress specifier for clinical outcomes in a large cohort of patients with major depressive disorder. J Clin Psychiatry. 2017;78(2):207-213. PubMed CrossRef
2. Zimmerman M, Clark H, McGonigal P, et al. Reliability and validity of the DSM-5 Anxious Distress Specifier Interview. Compr Psychiatry. 2017;76:11-17. PubMed CrossRef
3. Fava M, Rush AJ, Alpert JE, et al. What clinical and symptom features and comorbid disorders characterize outpatients with anxious major depressive disorder: a replication and extension. Can J Psychiatry. 2006;51(13):823-835. PubMed CrossRef
4. American Psychiatric Association. Diagnostic and Statistical Manual for Mental Disorders. Fifth Edition. Washington, DC: American Psychiatric Association; 2013.
5. Regier DA, Kuhl EA, Kupfer DJ. The DSM-5: Classification and criteria changes. World Psychiatry. 2013;12(2):92-98. PubMed CrossRef
6. Ionescu DF, Niciu MJ, Richards EM, et al. Pharmacologic treatment of dimensional anxious depression: a review. Prim Care Companion CNS Disord. 2014;16(3):10.4088/PCC.13r01621. PubMed CrossRef
7. McIntyre RS, Weiller E, Zhang P, et al. Brexpiprazole as adjunctive treatment of major depressive disorder with anxious distress: results from a post-hoc analysis of two randomised controlled trials. J Affect Disord. 2016;201:116-123. PubMed CrossRef
8. Zimmerman M, Chelminski I, Young D, et al. A clinically useful self-report measure of the DSM-5 anxious distress specifier for major depressive disorder. J Clin Psychiatry. 2014;75(6):601-607. PubMed CrossRef
9. Kessler RC, Sampson NA, Berglund P, et al. Anxious and non-anxious major depressive disorder in the World Health Organization World Mental Health Surveys. Epidemiol Psychiatr Sci. 2015;24(03):210-226. PubMed CrossRef
10. Lamers F, van Oppen P, Comijs HC, et al. Comorbidity patterns of anxiety and depressive disorders in a large cohort study: the Netherlands Study of Depression and Anxiety (NESDA). J Clin Psychiatry. 2011;72(3):341-348. PubMed CrossRef
11. Chan HN, Rush AJ, Nierenberg AA, et al. Correlates and outcomes of depressed out-patients with greater and fewer anxious symptoms: a CO-MED report. Int J Neuropsychopharmacol. 2012;15(10):1387-1399. PubMed CrossRef
12. Fava M, Alpert JE, Carmin CN, et al. Clinical correlates and symptom patterns of anxious depression among patients with major depressive disorder in STAR*D. Psychol Med. 2004;34(7):1299-1308. PubMed CrossRef
13. Fava M, Rush AJ, Alpert JE, et al. Difference in treatment outcome in outpatients with anxious versus nonanxious depression: a STAR*D report. Am J Psychiatry. 2008;165(3):342-351. PubMed CrossRef
14. Gaspersz R, Lamers F, Kent JM, et al. Anxious distress predicts subsequent treatment outcome and side effects in depressed patients starting antidepressant treatment. J Psychiatr Res. 2017;84:41-48. PubMed CrossRef
15. Wiethoff K, Bauer M, Baghai TC, et al. Prevalence and treatment outcome in anxious versus nonanxious depression: results from the German Algorithm Project. J Clin Psychiatry. 2010;71(8):1047-1054. PubMed CrossRef
16. Hovens JGFM, Giltay EJ, Spinhoven P, et al. Impact of childhood life events and childhood trauma on the onset and recurrence of depressive and anxiety disorders. J Clin Psychiatry. 2015;76(7):931-938. PubMed CrossRef
17. Hovens JGFM, Giltay EJ, Wiersma JE, et al. Impact of childhood life events and trauma on the course of depressive and anxiety disorders. Acta Psychiatr Scand. 2012;126(3):198-207. PubMed CrossRef
18. Trivedi MH, Rush AJ, Wisniewski SR, et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry. 2006;163(1):28-40. PubMed CrossRef
19. Romera I, Pérez V, Ciudad A, et al. Residual symptoms and functioning in depression, does the type of residual symptom matter? a post-hoc analysis. BMC Psychiatry. 2013;13(1):51. PubMed CrossRef
20. Weissman MM, Pilowsky DJ, Wickramaratne PJ, et al. Remissions in maternal depression and child psychopathology: a STAR*D-Child report. JAMA. 2006;295(12):1389-1398. PubMed CrossRef
21. Fifer SK, Mathias SD, Patrick DL, et al. Untreated anxiety among adult primary care patients in a health maintenance organization. Arch Gen Psychiatry. 1994;51(9):740-750. PubMed CrossRef
22. Krueger PM, Tran MK, Hummer RA, et al. Mortality attributable to low levels of education in the United States. PLoS One. 2015;10(7):e0131809. PubMed CrossRef
23. Roth T, Jaeger S, Jin R, et al. Sleep problems, comorbid mental disorders, and role functioning in the National Comorbidity Survey Replication (NCS-R). Biol Psychiatry. 2006;60(12):1364-1371. PubMed CrossRef
24. Bernert RA, Hom MA, Iwata NG, et al. Objectively assessed sleep variability as an acute warning sign of suicidal ideation in a longitudinal evaluation of young adults at high suicide risk. J Clin Psychiatry. 2017;78(6):e678-e687. PubMed CrossRef
25. Gelenberg AJ, Freeman MP, Markowitz JC, et al. Practice Guideline for the Treatment of Patients With Major Depressive Disorder Third Edition. Am J Psychiatry. 2010;167(10):1-152.
26. Lichtman JH, Bigger JT, Blumenthal JA, et al. Major depressive disorder and bipolar disorder predispose youth to accelerated atherosclerosis and early cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2008;118(17):1768-1775. PubMed CrossRef
27. Van der Kooy K, van Hout H, Marwijk H, et al. Depression and the risk for cardiovascular diseases: systematic review and meta analysis. Int J Geriatr Psychiatry. 2007;22(7):613-626. PubMed CrossRef
28. Almas A, Forsell Y, Iqbal R, et al. Severity of depression, anxious distress and the risk of cardiovascular disease in a Swedish population-based cohort. PLoS One. 2015;10(10):e0140742. PubMed CrossRef
29. Topp CW, טstergaard SD, S׸ndergaard S, et al. The WHO-5 Well-Being Index: a systematic review of the literature. Psychother Psychosom. 2015;84(3):167-176. PubMed CrossRef
30. Kroenke K, Spitzer RL, Williams JBW. The PHQ-9. J Gen Intern Med. 2001;16(9):606-613. PubMed CrossRef
31. Katzelnick DJ, Duffy FF, Chung H, et al. Depression outcomes in psychiatric clinical practice: using a self-rated measure of depression severity. Psychiatr Serv. 2011;62(8):929-935. PubMed CrossRef
32. Spitzer RL, Kroenke K, Williams JBW, et al. A brief measure for assessing generalized anxiety disorder: The GAD-7. Arch Intern Med. 2006;166(10):1092-1097. PubMed CrossRef
33. VitalSign6: Dallas Depression Center—UT Southwestern, Dallas, Texas. http://www.utsouthwestern.edu/education/medical-school/departments/psychiatry/divisions/depression/center/vital-sign6/. Accessed June 28, 2017.
34. Trombello J, South C, Cecil A, et al. Efficacy of a behavioral activation teletherapy intervention to treat depression and anxiety in primary care VitalSign6 program. Prim Care Companion CNS Disord. 2017; in press.
35. Guina J, Rossetter SR. DeRHODES BJ, et al. Benzodiazepines for PTSD: a systematic review and meta-analysis. J Psychiatr Pract. 2015;21(4):281-303. PubMed CrossRef
36. Kennedy SH, Lam RW, McIntyre RS, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder, section 3: pharmacological treatments. Can J Psychiatry. 2016;61(9):540-560. PubMed CrossRef
37. Schaffer A, McIntosh D, Goldstein BI, et al. The Canadian Network for Mood and Anxiety Treatments (CANMAT) task force recommendations for the management of patients with mood disorders and comorbid anxiety disorders. Ann Clin Psychiatry. 2012;24(1):6-22. PubMed
38. Ionescu DF, Niciu MJ, Mathews DC, et al. Neurobiology of anxious depression: a review. Depress Anxiety. 2013;30(4):374-385. PubMed CrossRef
39. Fava M, Rosenbaum JF, Hoog SL, et al. Fluoxetine versus sertraline and paroxetine in major depression: tolerability and efficacy in anxious depression. J Affect Disord. 2000;59(2):119-126. PubMed CrossRef
40. Thaler KJ, Morgan LC, Van Noord M, et al. Comparative effectiveness of second-generation antidepressants for accompanying anxiety, insomnia, and pain in depressed patients: a systematic review. Depress Anxiety. 2012;29(6):495-505. PubMed CrossRef
41. Davidson JRT, Meoni P, Haudiquet V, et al. Achieving remission with venlafaxine and fluoxetine in major depression: its relationship to anxiety symptoms. Depress Anxiety. 2002;16(1):4-13. PubMed CrossRef
42. Sir A, D’ Souza RF, Uguz S, et al. Randomized trial of sertraline versus venlafaxine XR in major depression: efficacy and discontinuation symptoms. J Clin Psychiatry. 2005;66(10):1312-1320. PubMed CrossRef
43. Mohamed S, Johnson GR, Chen P, et al. Effect of antidepressant switching vs augmentation on remission among patients with major depressive disorder unresponsive to antidepressant treatment: the VAST-D randomized clinical trial. JAMA. 2017;318(2):132-145. PubMed CrossRef
44. Trivedi MH, Rush AJ, Carmody TJ, et al. Do bupropion SR and sertraline differ in their effects on anxiety in depressed patients? J Clin Psychiatry. 2001;62(10):776-781. PubMed CrossRef
45. Rush AJ, Trivedi MH, Stewart JW, et al. Combining Medications to Enhance Depression Outcomes (CO-MED): acute and long-term outcomes of a single-blind randomized study. Am J Psychiatry. 2011;168(7):689-701. PubMed CrossRef
46. Thase ME, Friedman ES, Biggs MM, et al. Cognitive therapy versus medication in augmentation and switch strategies as second-step treatments: a STAR*D report. Am J Psychiatry. 2007;164(5):739-752. PubMed CrossRef
47. Farabaugh A, Alpert J, Wisniewski SR, et al. Cognitive therapy for anxious depression in STAR(*) D: what have we learned? J Affect Disord. 2012;142(1-3):213-218. PubMed CrossRef
48. Trivedi MH, Greer TL, Church TS, et al. Exercise as an augmentation treatment for nonremitted major depressive disorder: a randomized, parallel dose comparison. J Clin Psychiatry. 2011;72(5):677-684. PubMed CrossRef
49. Schröder CM, O’ Hara R. Depression and obstructive sleep apnea (OSA). Ann Gen Psychiatry. 2005;4(1):13. PubMed CrossRef
50. Nelson JC, Papakostas GI. Atypical antipsychotic augmentation in major depressive disorder: a meta-analysis of placebo-controlled randomized trials. Am J Psychiatry. 2009;166(9):980-991. PubMed CrossRef
51. Spielmans GI, Berman MI, Linardatos E, et al. Adjunctive atypical antipsychotic treatment for major depressive disorder: a meta-analysis of depression, quality of life, and safety outcomes. PLoS Med. 2013;10(3):e1001403. PubMed CrossRef
52. Trivedi MH, Thase ME, Fava M, et al. Adjunctive aripiprazole in major depressive disorder: analysis of efficacy and safety in patients with anxious and atypical features. J Clin Psychiatry. 2008;69(12):1928-1936. PubMed CrossRef
53. Pae C-U, Forbes A, Patkar AA. Aripiprazole as adjunctive therapy for patients with major depressive disorder: overview and implications of clinical trial data. CNS Drugs. 2011;25(2):109-127. PubMed CrossRef
54. Hobart M, Skuban A, Zhang P, et al. A randomized, double-blind, placebo-controlled study of brexpiprazole as adjunctive therapy in the treatment of adults with major depressive disorder. ACNP 55th Annual Meeting. December 4-8, 2016; Hollywood, FL.
55. Thase ME, Youakim JM, Skuban A, et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebo-controlled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 2015;76(9):1224-1231. PubMed CrossRef
56. Bandelow B, Bauer M, Vieta E, et al. Extended release quetiapine fumarate as adjunct to antidepressant therapy in patients with major depressive disorder: pooled analyses of data in patients with anxious depression versus low levels of anxiety at baseline. World J Biol Psychiatry. 2014;15(2):155-166. PubMed CrossRef
57. Cha DS, McIntyre RS. Treatment-emergent adverse events associated with atypical antipsychotics. Expert Opin Pharmacother. 2012;13(11):1587-1598. PubMed CrossRef
58. Corya SA, Williamson D, Sanger TM, et al. A randomized, double-blind comparison of olanzapine/fluoxetine combination, olanzapine, fluoxetine, and venlafaxine in treatment-resistant depression. Depress Anxiety. 2006;23(6):364-372. PubMed CrossRef
59. Thase ME, Corya SA, Osuntokun O, et al. A randomized, double-blind comparison of olanzapine/fluoxetine combination, olanzapine, and fluoxetine in treatment-resistant major depressive disorder. J Clin Psychiatry. 2007;68(2):224-236. PubMed CrossRef
60. Symbyax [package insert]. Indianapolis, IN: Eli Lilly and Company; 2017.
61. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Engl J Med. 2006;354(12):1243-1252. PubMed CrossRef
62. Buspirone hydrochloride [package insert]. Corona, CA: Watson Laboratories; 2013.
63. Thase ME. Update on partial response in depression. J Clin Psychiatry. 2009;70(suppl 6):4-9. PubMed CrossRef
64. Gijsman H. Review: antidepressants plus benzodiazepines lead to fewer dropouts and less depression severity at 4 weeks in major depression. Evid Based Ment Health. 2001;4(2):45. CrossRef
65. Jones CM, Mack KA, Paulozzi LJ. Pharmaceutical overdose deaths, United States, 2010. JAMA. 2013;309(7):657-659. PubMed CrossRef
66. Davis MA, Lin LA, Liu H, et al. Prescription opioid use among adults with mental health disorders in the United States. J Am Board Fam Med. 2017;30(4):407-417. PubMed CrossRef
67. Cooper-Kazaz R, Lerer B. Efficacy and safety of triiodothyronine supplementation in patients with major depressive disorder treated with specific serotonin reuptake inhibitors. Int J Neuropsychopharmacol. 2008;11(05). PubMed CrossRef
68. Crossley NA, Bauer M. Acceleration and augmentation of antidepressants with lithium for depressive disorders: two meta-analyses of randomized, placebo-controlled trials. J Clin Psychiatry. 2007;68(6):935-940. PubMed CrossRef
69. Papakostas GI, Shelton RC, Zajecka JM, et al. L-methylfolate as adjunctive therapy for SSRI-resistant major depression: results of two randomized, double-blind, parallel-sequential trials. Am J Psychiatry. 2012;169(12):1267-1274. PubMed CrossRef
70. Fava M, Schaefer K, Huang H, et al. A post hoc analysis of the effect of nightly administration of eszopiclone and a selective serotonin reuptake inhibitor in patients with insomnia and anxious depression. J Clin Psychiatry. 2011;72(4):473-479. PubMed CrossRef
71. World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines. Geneva, Switzerland: World Health Organization; 1992.
72. Trivedi MH. Tools and strategies for ongoing assessment of depression: a measurement-based approach to remission. J Clin Psychiatry. 2009;70(suppl 6):26-31. PubMed CrossRef
73. Patten SB, Kennedy SH, Lam RW, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) clinical guidelines for the management of major depressive disorder in adults, i: classification, burden and principles of management. J Affect Disord. 2009;117:S5-S14. PubMed CrossRef
74. McIntyre RS. Florida best practice psychotherapeutic medication guidelines for adults with major depressive disorder. J Clin Psychiatry. 2017;78(6):703-713. PubMed CrossRef
75. Gollan JK, Fava M, Kurian B, et al. What are the clinical implications of new onset or worsening anxiety during the first two weeks of SSRI treatment for depression? Depress Anxiety. 2012;29(2):94-101. PubMed CrossRef
*Cases have been fictionalized and were provided by the meeting participants to illustrate material discussed in the article.
This PDF is free for all visitors!