Primary care clinicians use urine drug testing to monitor compliance in patients prescribed controlled substances. A negative test for the prescribed medication can suggest diversion to family or friends or illicit sale of the medication. Despite not being recommended for this use, qualitative immunoassay screens are frequently used to guide decision making. Urine samples that screen negative are frequently discarded by the laboratory before definitive testing can be performed.
Case Report
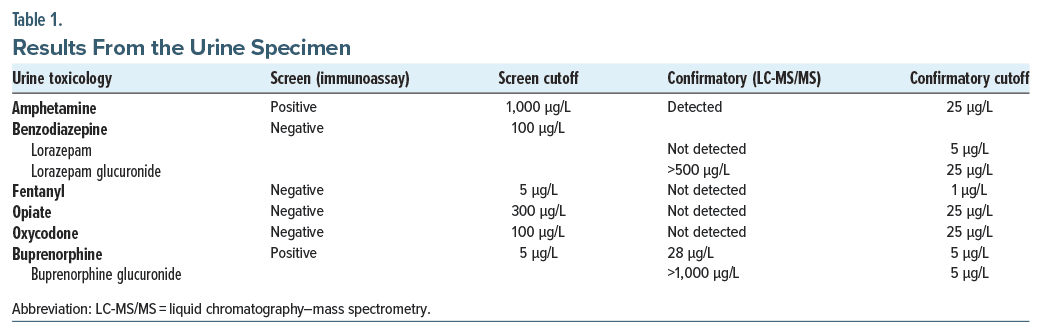
Our laboratory was consulted by a primary care provider working in an outpatient pain management clinic regarding the results of a urine toxicology immunoassay screening panel (Table 1). The urine specimen was from a 43-year-old man with a history of opioid use disorder, now in sustained remission, being treated for anxiety, attention-deficit/hyperactivity disorder, and chronic pain. His daily medications included buprenorphine-naloxone (8-2 mg tablet), dextroamphetamine amphetamine (30 mg), and lorazepam (0.5 mg).
The screening results were consistent with those of several prior specimens in the past year. The provider was not concerned about a relapse, as the opiate screen was negative, and the buprenorphine screen was positive. However, in the context of an ongoing shortage of lorazepam, the persistently negative benzodiazepine results were concerning for diversion (eg, selling prescription for profit). When confronted about the possibility of diverting lorazepam, the patient became agitated and defensive and accused someone of substituting his urine. This led to distrust and a disruption in the alliance with his interdisciplinary care team.
After consulting with the laboratory, the urine sample was sent for testing by liquid chromatography–mass spectrometry (LC-MS/MS), a more sensitive and specific methodology (Table 1). Although the parent drug was not detected, the primary metabolite found in urine, lorazepam glucuronide, was present in high concentrations (>500 μg/L), indicating a false-negative immunoassay screen and compliance with his prescription.
Discussion
Immunoassays are not intended to monitor adherence to benzodiazepines or other medications.1,2 The most widely used commercial urine benzodiazepine immunoassays have poor cross-reactivity for lorazepam and its glucuronidated metabolite.3–5 Further, studies investigating the performance of point of care assays have shown a high false-negative rate (50%) for lorazepam.6 The sensitivity of benzodiazepine immunoassays can be improved by pretreating the specimen with β-glucuronidase. This hydrolyzes the glucuronide conjugate, releases the active form of the benzodiazepine (eg, lorazepam), and makes it detectable by the immunoassay.3–5 However, pretreatment is operationally challenging for laboratories and increases turnaround time. Further, any alterations to a US Food and Drug Administration (FDA)–cleared/ approved assay revert it to a high complexity test, necessitating validation of the modification and more rigorous regulatory requirements. A recent FDA ruling, if not overturned, will effectively prohibit such modifications by individual labs.7
Most providers are aware that immunoassays have limitations. The most obvious drawback is specificity: benzodiazepine immunoassays cannot determine which benzodiazepines are present. In this case, however, sensitivity is also poor. The provider consulted the manufacturer’s package insert for the benzodiazepine immunoassay, which lists 62% cross reactivity with lorazepam.8 However, this is misleading, as lorazepam is eliminated primarily as the glucuronide in urine,9 and the cross reactivity with this metabolite is negligible (0.50%). It would require approximately 20,000 μg/L of lorazepam glucuronide, a very high and even potentially fatal concentration, to trigger a positive screening result. In fact, a patient recently presenting to our hospital with a lorazepam overdose was negative by immunoassay but confirmed positive by LC-MS/MS. Newer immunoassays have better sensitivity for lorazepam use but still have a high rate of false negatives, particularly in specimens where only the glucuronide is present.10,11
Routinely using direct-to-definitive testing by LC-MS/MS can be more costly. However, it should be considered when monitoring patients for compliance with benzodiazepine prescriptions, especially in the case of lorazepam.
Article Information
Published Online: April 3, 2025. https://doi.org/10.4088/PCC.24cr03889
© 2025 Physicians Postgraduate Press, Inc.
Prim Care Companion CNS Disord 2025;27(2):24cr03889
Submitted: November 8, 2024; accepted January 27, 2025.
To Cite: Uljon S, Donnelly-Morell ML, Tolan N, et al. Is my patient diverting their lorazepam? Prim Care Companion CNS Disord 2025;27(2):24cr03889.
Author Affiliations: Department of Pathology, Massachusetts General Hospital, Mass General Brigham, Boston, Massachusetts (Uljon, Donnelly-Morell); Department of Pathology, Brigham and Women’s Hospital, Mass General Brigham, Boston, Massachusetts (Tolan, Melanson).
Corresponding Author: Sacha Uljon, MD, PhD, Core Laboratory, Department of Pathology, Massachusetts General Hospital, 55 Fruit St, GRB 554, Boston, MA 02114- 2696 ([email protected]).
Relevant Financial Relationships: None.
Funding/Support: None.
Additional Information: Information has been been de-identified to protect patient anonymity.
References (11)

- Jannetto PJ, Bratanow NC, Clark WA, et al. Executive summary: American association of clinical chemistry laboratory medicine practice guideline-using clinical laboratory tests to monitor drug therapy in pain management patients. J Appl Lab Med. 2018;2(4):489–526. CrossRef
- Melanson SE, Ptolemy AS, Wasan AD. Optimizing urine drug testing for monitoring medication compliance in pain management. Pain Med. 2013;14(12):1813–1820. PubMed CrossRef
- Darragh A, Snyder ML, Ptolemy AS, et al. KIMS, CEDIA, and HS-CEDIA immunoassays are inadequately sensitive for detection of benzodiazepines in urine from patients treated for chronic pain. Pain Physician. 2014;17(4):359–366. PubMed
- Lund K, Menlyadiev M, Lee K, et al. Comparison of two highly sensitive benzodiazepine immunoassay lab developed tests for urine drug testing in clinical specimens. J Mass Spectrom Adv Clin Lab. 2023;28:91–98. CrossRef
- Rossi B, Freni F, Vignali C, et al. Comparison of two immunoassay screening methods and a LC-MS/MS in detecting traditional and designer benzodiazepines in urine. Molecules. 2021;27(1):112. CrossRef
- Mikel C, Pesce AJ, Rosenthal M, et al. Therapeutic monitoring of benzodiazepines in the management of pain: current limitations of point of care immunoassays suggest testing by mass spectrometry to assure accuracy and improve patient safety. Clin Chim Acta. 2012;413(15–16):1199–1202. PubMed CrossRef
- Office of the Federal Register, National Archives and Records Administration. 89 FR 37286 - Medical Devices; Laboratory Developed Tests [Government]. Office of the Federal Register, National Archives and Records Administration; 2024. https://www.govinfo.gov/app/details/FR-2024-05-06/2024-08935
- Roche Benzodiazepines Plus (BNZ) assay [package insert]. Roche Diagnostics; 2020.
- Elliott HW. Metabolism of lorazepam. Br J Anaesth. 1976;48(10):1017–1023. PubMed CrossRef
- Ge M, Alabi A, Kelner MJ, et al. Evaluation of a benzodiazepine immunoassay for urine drug testing in clinical specimens. J Appl Lab Med. 2024;9(6):964–976. CrossRef
- Tolan NV, Uljon S, Lauren Donnelly-Morell M, et al. Despite the improved clinical sensitivity of the Roche benzodiazepines II assay it cannot replace mass spectrometry in all patient populations. J Mass Spectrom Adv Clin Lab. 2024;33:14–20.
This PDF is free for all visitors!